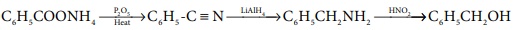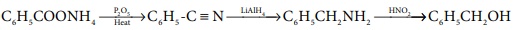Chemistry - Online Test
Q1. If one strand of the DNA has the sequence ‘ATGCTTGA’, then the sequence of complementary strand would be
Answer : Option A
Explaination / Solution:
No Explaination.
Q2. In a homogeneous reaction A →B + C + D , the initial pressure
was P0 and after time t it was P.
expression for rate constant in terms of P0,
P and t will be
Answer : Option A
Explaination / Solution:


Q3. The hydrogen ion concentration of a buffer solution consisting of a weak acid and its salts is given by
Answer : Option A
Explaination / Solution:
According to Henderson equation
pH = pKa + log ([salt]/[acid])
ie. - log [H+ ] = -
log Ka +log ([salt] / [acid])
-log[H+] = log ([salt]/ [acid]) ×
(1/Ka)
log (1/[H+]) = log ([salt]/ [acid]) × (1/Ka)
∴[H+] = Ka
{[acid] / [salt] }
Q4. In the electrolytic refining of copper, which one of the following is used as anode?
Answer : Option B
Explaination / Solution:
No Explaination.
Q5.
Consider the change in oxidation state of Bromine corresponding to
different emf values as shown in the diagram below:
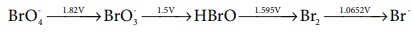
Then the species undergoing disproportionation is
Answer : Option D
Explaination / Solution:
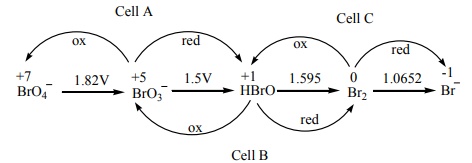
( Ecell )A =-1.82+1.5=-0.32V
( Ecell )B =-1.5+1.595=+0.095V
( Ecell )c =-1.595+1.0652=-0.529V
The species undergoing
disproportionation is HBrO
Q6.
For the cell reaction
2Fe3+ (aq) + 2l−(aq) → 2Fe2+ (aq) + l2 (aq)
Answer : Option A
Explaination / Solution:
No Explaination.
Q7. On reacting with neutral ferric chloride, phenol gives
Answer : Option B
Explaination / Solution:
No Explaination.
Q8.
Assertion : Ce4+ is used as an oxidizing agent in
volumetric analysis.
Answer : Option A
Explaination / Solution:
No Explaination.
Q9. In which of the following reactions new carbon – carbon bond is not formed?
Answer : Option D
Explaination / Solution:
No Explaination.
Q10. Ammonium
salt of benzoic acid is heated strongly with P2 O5 and
the product so formed is reduced and then treated with NaNO2 / HCl
at low temperature. The final compound formed is
Answer : Option B
Explaination / Solution: