Chemistry - Online Test
Q1. One
mole of an organic compound (A) with the formula C3 H8O
reacts completely with two moles of HI to form X and Y. When Y is boiled with
aqueous alkali it forms Z. Z answers the iodoform test. The compound (A) is
Answer : Option D
Explaination / Solution:
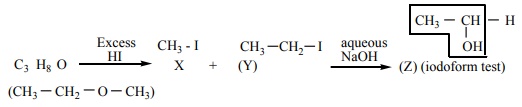
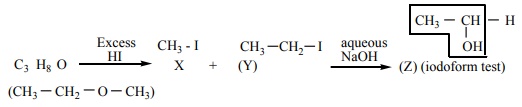
Q2. Which one of the following statements related to lanthanons is incorrect?
Answer : Option C
Explaination / Solution:
As we move from La to Lu , their metallic behaviour because almost similar to that of aluminium.
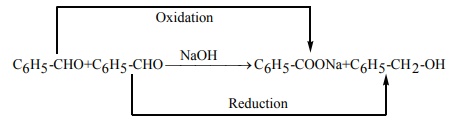
Q3. Which one of the following undergoes reaction with 50% sodium hydroxide solution to give the corresponding alcohol and acid
Answer : Option A
Explaination / Solution:

Q4. Fac-mer isomerism is shown by
Answer : Option C
Explaination / Solution:
[Co (NH3)3(Cl)3]
Q5. When 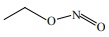 is reduced with Sn / HCl the pair of compounds formed are
is reduced with Sn / HCl the pair of compounds formed are
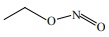 is reduced with Sn / HCl the pair of compounds formed are
is reduced with Sn / HCl the pair of compounds formed are
Answer : Option A
Explaination / Solution:
No Explaination.
Q6. If ‘a’ is the length of the side of the cube, the distance between the body centered atom and one corner atom in the cube will be
Answer : Option D
Explaination / Solution:
if a is the length of the side, then the length of the leading
diagonal passing through the body centered atom is √3a
Required distance (√3/2) a
Q7. Which of the following statement is not correct?
Answer : Option C
Explaination / Solution:
No Explaination.
Q8. During the decomposition of
H2O2 to give dioxygen, 48 g O2 is formed per
minute at certain point of time. The rate of formation of water at this point
is
Answer : Option D
Explaination / Solution:
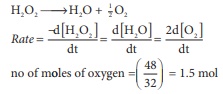
no of moles of oxygen = (48/32) =
1.5 mol
∴
rate of formation of oxygen = 2 ×1.5
= 3 mol min-1
Q9. Which of the following can act as Lowry – Bronsted acid as well as base?
Answer : Option C
Explaination / Solution:
HPO42− can have the ability to accept a proton to form H2PO−4.
It can also have the ability to
donate a proton to form PO4-3
Q10. Which of the following is used for concentrating ore in metallurgy?
Answer : Option D
Explaination / Solution:
No Explaination.
