Chemistry - Online Test
Q1. Which one of the following is most basic?
Answer : Option B
Explaination / Solution:
CH3
is a+I group, all other – I group. +I group increase the electron density on NH2
and hence increases the basic nature.
Q2. if ‘a’ stands for the edge length of the cubic system ; sc , bcc, and fcc. Then the ratio of radii of spheres in these systems will be respectively.
Answer : Option C
Explaination / Solution:
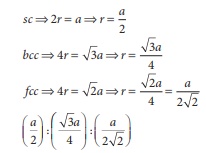
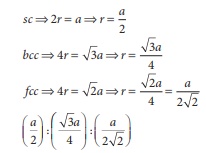
Q3. Complete hydrolysis of cellulose gives
Answer : Option D
Explaination / Solution:
No Explaination.
Q4. For the reaction N2O5(g) → 2NO2
(g) + ½ O2(g), the value of rate of disappearance of N2O5
is given as 6.5 × 10-2 mol L-1
s-1. The rate of formation of NO2 and O2 is
given respectively as
Answer : Option C
Explaination / Solution:
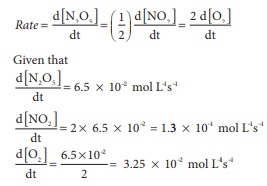
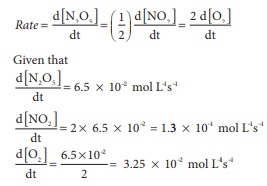
Q5. H2PO4-
the conjugate base of
Answer : Option C
Explaination / Solution:
H3PO4
+H − OH ↔ H3O+ + H2PO4-
acid 1 +
base 1 ↔ acid 2 + base 2
∴H2PO4− is the conjugate base of H3PO4
Q6. The following set of
reactions are used in refining Zirconium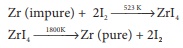
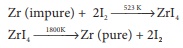
This method is known as
Answer : Option B
Explaination / Solution:
No Explaination.
Q7. A
conductivity cell has been calibrated with a 0.01M, 1:1 electrolytic solution
(specific conductance ( κ =1.25 ×10−3 S cm−1 ) in the cell and the measured resistance was 800 W at 25º C . The cell constant is,
Answer : Option C
Explaination / Solution:
R = ρ. l/A
cell constant = R/ρ
= κ.R (1/ ρ =κ )
= 1.25 × 10−3 Ω−1cm−1
× 800 Ω
= 1 cm−1
Q8. The compound that is used in nuclear reactors as protective shields and control rods is
Answer : Option A
Explaination / Solution:
No Explaination.
Q9. On which of the following properties does the coagulating power of an ion depend ?
Answer : Option A
Explaination / Solution:
No Explaination.
Q10. Among the following the correct order of acidity is
Answer : Option D
Explaination / Solution:
No Explaination.
