Chemistry - Online Test
Q1. The
pH of 10-5M KOH solution will be
Answer : Option A
Explaination / Solution:
KOH →
K+ + OH-
10-5m 10-5m 10-5m
KOH (10-5m) → K+
(10-5m) + OH- (10-5m)
[OH-] = 10-5M.
pH= 14 - pOH
pH = 14 - ( - log [OH-]
)
= 14 + log [OH- ]
= 14 + log10−5
= 14−5
= 9.
Q2. Considering Ellingham diagram, which of the following metals can be used to reduce alumina?
Answer : Option C
Explaination / Solution:
No Explaination.
Q3.
17. The equivalent conductance of M/36 solution of a weak monobasic acid
is 6 mho cm2 equivalent –1 and at infinite dilution is
400 mho cm2 equivalent –1. The dissociation constant of
this acid is
Answer : Option B
Explaination / Solution:
α = Λ / Λo = 6/400
Ka =α2C
= 6/400 × 6/400 × 1/36
= 6.25 ×10−6
Q4. Thermodynamically the most stable form of carbon is
Answer : Option B
Explaination / Solution:
No Explaination.
Q5. If x is the amount of adsorbate and m is the amount of adsorbent, which of the following relations is not related to adsorption process?
Answer : Option D
Explaination / Solution:
No Explaination.
Q6. Which one of the following orders is correct for the bond dissociation enthalpy of halogen molecules?
Answer : Option D
Explaination / Solution:
No Explaination.
Q7.
The reactions
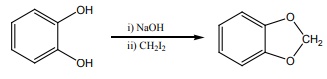
Answer : Option C
Explaination / Solution:
No Explaination.
Q8. When a brown compound of Mn
(A) ids treated with HCl , it gives a gas (B) . The gas (B) taken in excess
reacts with NH3 to give an explosive compound (C). The compound A, B
and C are
Answer : Option A
Explaination / Solution:
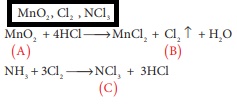
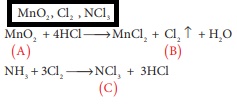
Q9. Which one of the following reaction is an example of disproportionation reaction
Answer : Option B
Explaination / Solution:
No Explaination.
Q10. Which of the following is paramagnetic in nature?
Answer : Option C
Explaination / Solution:
(a) Zn2+ (d10
⟹
diamagnetic)
(b) Co3+ (d6
Low spin ⟹ t2g6, eg0
diamagnetic)
(c) Ni2+ (d8
Low spin ⟹ t2g6, eg2
paramagnetic)
(d) [Ni(CN)4]2-
(dsp2 ; square planar, diamagnetic)
