Chemistry - Online Test
Q1. Adsorption of a gas on solid metal surface is spontaneous and exothermic, then
Answer : Option D
Explaination / Solution:
∆S is –ve
Q2. Which of the following is strongest acid among all?
Answer : Option A
Explaination / Solution:
No Explaination.
Q3. Which of the following compound can be used as artifreeze in automobile radiators?
Answer : Option D
Explaination / Solution:
No Explaination.
Q4. The number of moles of
acidified KMnO4 required to oxidize 1 mole of ferrous oxalate(FeC2O4)
is
Answer : Option C
Explaination / Solution:


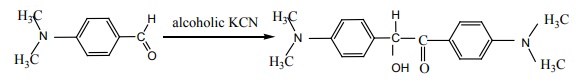
Q5.
Assertion : p – N, N – dimethyl aminobenzaldehyde
undergoes benzoin condensation
Answer : Option B
Explaination / Solution:

Q6. Formula of tris(ethane-1,2-diamine)iron(II)phosphate
Answer : Option D
Explaination / Solution:
[Fe (en )3]2+
(PO43-)
Q7. Which of the following amines does not undergo acetylation?
Answer : Option D
Explaination / Solution:
triethyl amine ( 3 amine)
Q8. The yellow colour in NaCl crystal is due to
Answer : Option A
Explaination / Solution:
No Explaination.
Q9. Which of the following vitamins is water soluble?
Answer : Option D
Explaination / Solution:
No Explaination.
Q10. The rate constant of a reaction
is 5.8 × 10−2
s−1 . The order of the reaction is
Answer : Option A
Explaination / Solution:
The unit of rate constant is s-1 and it indicates that the reaction is first order.
