Chemistry - Online Test
Q1. In which case chiral carbon is not generated by reaction with HCN
Answer : Option A
Explaination / Solution:
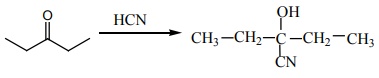

Q2. A complex in which the oxidation number of the metal is zero is
Answer : Option C
Explaination / Solution:
(a) Fe2+
(b) Fe3+
(c) Fe0
Q3. Secondary nitro alkanes react with nitrous acid to form
Answer : Option B
Explaination / Solution:
No Explaination.
Q4. The fraction of total volume occupied by the atoms in a simple cubic is
Answer : Option B
Explaination / Solution:
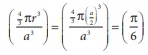
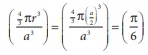
Q5. The secondary structure of a protein refers to
Answer : Option D
Explaination / Solution:
No Explaination.
Q6.
Assertion: rate of reaction doubles
when the concentration of the reactant is doubles if it is a first order
reaction.
Answer : Option C
Explaination / Solution:
For a first reaction, If
the concentration of reactant is doubled, then the rate of reaction also
doubled.
Rate constant is
independent of concentration and is a constant at a constant temperature,
Q7. The
dissociation constant of a weak acid is 1 ×10-3.
In order to prepare a buffer solution with a pH = 4, the [Acid]/[Salt] ratio
should be
Answer : Option D
Explaination / Solution:
K a =1 ×10-3
pH=4
[salt] /
[Acid] = ?
pH = pKa +log {[Salt]/[Acid]}
4 = - log10 (1 ×10-3 ) + log {[Salt]/ [Acid]}
4 = 3 + log ([Salt]/ [Acid])
1= log10 ([Salt]/[Acid])
[Salt] / [Acid] =101
i.e., [Salt] / [Acid] = 1 / 10
1:10
Q8. Extraction of gold and silver involves leaching with cyanide ion. silver is later recovered by
Answer : Option C
Explaination / Solution:
No Explaination.
Q9. In
H2 -O2 fuel cell the reaction occurs at cathode is
Answer : Option A
Explaination / Solution:
No Explaination.
Q10. Duralumin is an alloy of
Answer : Option D
Explaination / Solution:
Al-95% , Cu-4% , Mn-0.5% , Mn-0.5%
