Chemistry - Online Test
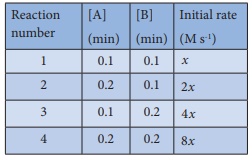
Predict the rate law of
the following reaction based on the
data given below
2A + B → C + 3D
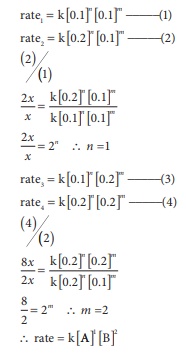
x ml of 0.1 M NaOH + x ml of 0.01 M HCl
No of moles of NaOH = 0.1 × x × 10–3 = 0.1x
× 10-3
No of moles of HCl = 0.01 × x × 10-3 = 0.01x × 10-3
No of moles of NaOH after mixing
= 0.1x × 10-3 - 0.01x × 10-3
= 0.09x
× 10-3
Concentration of NaOH= [0.09x × 10−3 ] /
[2x × 10−3] = 0.045
[OH-] = 0.045
POH = −log (4.5 × 10−2
)
= 2 −log 4.5
= 2- 0.65 = 1.35
pH = 14-1.35=12.65
Assertion : pure iron when heated in dry air is
converted with a layer of rust.
Both are
false
i) Dry
air has no reaction with iron
ii) Rust
has the composition Fe2 O3 . x H2O
The coagulation values in millimoles per litre of the electrolytes used
for the coagulation of As2S3 are given below
(I) (NaCl)=52 (II) ((BaCl2 )=0.69 (III) (MgSO4
)=0.22
