Chemistry - Online Test
Q1. What is the activation
energy for a reaction if its rate doubles when the temperature is raised from
200K to 400K? (R = 8.314 JK-1mol-1)
Answer : Option C
Explaination / Solution:
T1 = 200K ; k = k1
T2 = 400K ; k = k2
= 2k1

Q2. Non stick cook wares generally have a coating of a polymer, whose monomer is
Answer : Option D
Explaination / Solution:
No Explaination.
Q3. Equal
volumes of three acid solutions of pH 1,2 and 3 are mixed in a vessel. What
will be the H+ ion concentration in the mixture?
Answer : Option A
Explaination / Solution:
pH = -log10[H+]
∴[H+]=10-pH
Let the volume be x mL
V1M1 + V2M2
+ V3M3 =VM
∴
x mL of 10-1M+ x mL of
10-2M + x mL of 10-3 M
= 3x mL of [H+]
∴[H+ ] = x[0.1+0.01+0.001] / 3x
= ( 0.1+ 0.01+ 0.001 ) / 3
= 0..111 / 3
= 0.037
= 3.7 ×10−2
Q4. Flux is a substance which is used to convert
Answer : Option B
Explaination / Solution:
No Explaination.
Q5. Which of the following electrolytic solution has the least specific conductance
Answer : Option B
Explaination / Solution:
In general, specific conductance of an electrolyte decreases with dilution. So, 0.002N solution has least specific conductance.
Q6. Which of the following is not sp2 hybridised?
Answer : Option D
Explaination / Solution:
dry ice – solid CO2 in which carbon
is in sp hybridized state
Q7. In
an electrical field, the particles of a colloidal system move towards cathode.
The coagulation of the same sol is studied using K2 SO4
(i), Na3 PO4 (ii),K4 [Fe(CN)6 ]
(iii) and NaCl (iv) Their coagulating power should be
Answer : Option B
Explaination / Solution:
No Explaination.
Q8. The correct order of the thermal stability of hydrogen halide is
Answer : Option B
Explaination / Solution:
No Explaination.
Q9. In
the reaction Ethanol 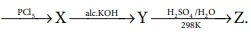 The ‘Z’ is
The ‘Z’ is
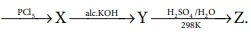 The ‘Z’ is
The ‘Z’ is
Answer : Option D
Explaination / Solution:
CH3 - CH2 -
OH ---PCl5→ CH3
- CH2 - Cl --ale.KOH→CH2 = CH2 --H2SO4/H2O→ CH3 - CH2 – OH
(Z) ethanol
Q10. Which of the following statements is not true?
Answer : Option B
Explaination / Solution:
No Explaination.
