Chemistry - Online Test
Q1. Which of these is not a monomer for a high molecular mass silicone polymer?
Answer : Option A
Explaination / Solution:
No Explaination.
Q2. The phenomenon observed when a beam of light is passed through a colloidal solution is
Answer : Option D
Explaination / Solution:
Tyndall effect-scattering of light
Q3. Among the following, which is the strongest oxidizing agent?
Answer : Option B
Explaination / Solution:
No Explaination.
Q4.
Assertion : Phenol is more acidic than ethanol
Answer : Option A
Explaination / Solution:
No Explaination.
Q5. In acid medium, potassium permanganate oxidizes oxalic acid to
Answer : Option B
Explaination / Solution:
5(COO)22-
+ 2MnO 4 + 16H+ → 2Mn2+ + 10CO2 ↑ + 8H2O
Q6. CH3Br
----KCN → (A) ---H3O+→(B) ----PCl5 →(C) product (C) is
Answer : Option A
Explaination / Solution:
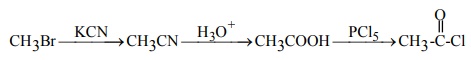
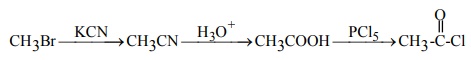
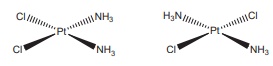
Q7. Which type of isomerism is
exhibited by Pt ( NH3)2 Cl2 ?
Answer : Option D
Explaination / Solution:

Q8. The order of basic strength for methyl substituted amines in aqueous solution is
Answer : Option D
Explaination / Solution:
No Explaination.
Q9. The ionic radii of A+
and B− are 0.98 × 10−10 m and 1.81 × 10−10 m . the coordination number of each ion in AB is
Answer : Option C
Explaination / Solution:
rC+/ rA-
= 0.98 ×10−10 / 1.81 ×10−10 = 0.54
it is in the range of 0.
414 - 0.732 , hence the coordination number of each ion is 6
Q10. Which one of the following is not produced by body?
Answer : Option D
Explaination / Solution:
No Explaination.
