Chemistry - Online Test
Q1. Which one of the following reduces tollens reagent
Answer : Option A
Explaination / Solution:
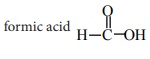
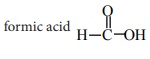
Q2. How many geometrical
isomers are possible for [ Pt(Py)(NH3)(Br)(Cl)] ?
Answer : Option A
Explaination / Solution:
Three isomers. If we
consider any one of the ligands as reference ( say Py), the arrangement of other three ligands ( NH3, Br
and Cl‑ ) with respect to (Py) gives three geometrical
isomers.
Q3. 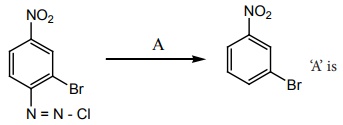

Answer : Option A
Explaination / Solution:
No Explaination.
Q4. CsCl has bcc arrangement, its unit cell edge length is 400pm, its inter atomic distance is
Answer : Option D
Explaination / Solution:
Solution
√3 a = rCs+ +
2rCl- + rCs+
(√3/2) a = ( rCl-
+ rCs+)
(√3/2) x 400 = inter
ionic distance
Q5. The number of sp2 and sp3 hybridised carbon in fructose are respectively
Answer : Option D
Explaination / Solution:
No Explaination.
Q6. 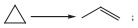 This reaction follows first
order kinetics. The rate constant at particular temperature is 2.303 × 10−2 hour−1 . The initial concentration of cyclopropane is 0.25M. What will be the concentration of cyclopropane after 1806 minutes?
(log 2 = 0.3010)
This reaction follows first
order kinetics. The rate constant at particular temperature is 2.303 × 10−2 hour−1 . The initial concentration of cyclopropane is 0.25M. What will be the concentration of cyclopropane after 1806 minutes?
(log 2 = 0.3010)
 This reaction follows first
order kinetics. The rate constant at particular temperature is 2.303 × 10−2 hour−1 . The initial concentration of cyclopropane is 0.25M. What will be the concentration of cyclopropane after 1806 minutes?
(log 2 = 0.3010)
This reaction follows first
order kinetics. The rate constant at particular temperature is 2.303 × 10−2 hour−1 . The initial concentration of cyclopropane is 0.25M. What will be the concentration of cyclopropane after 1806 minutes?
(log 2 = 0.3010)
Answer : Option B
Explaination / Solution:


Q7.
Assertion: 2-methyl-1,3-butadiene is the monomer of natural rubber
Answer : Option C
Explaination / Solution:
No Explaination.
Q8. The
solubility of AgCl (s) with solubility product 1.6 ×10-10 in 0.1M NaCl solution would be
Answer : Option B
Explaination / Solution:
AgCl(s) ↔ Ag+ (aq) + Cl-(aq)
NaCl (0.1 M) → Na+(0.1
M) + Cl- (0.1 M)
Ksp = 1..6 ×10-10
Ksp = [Ag+][Cl-]
Ksp = (s)(s+0.1)
0.1>>>s
∴
s + 0.1 = 0.1
∴
S = [ 1..6 ×10-10 ] / 0.1 = 1.6 ×10-9
Q9. Which one of the following ores is best concentrated by froth – floatation method?
Answer : Option C
Explaination / Solution:
No Explaination.
Q10. While charging lead storage battery
Answer : Option C
Explaination / Solution:
Charging : anode : PbSO4
(s)+ 2e− → Pb (s) +
SO4-2 (aq)
Cathode : PbSO4 (s)+
2H2 O (l) → PbO2 (s) + SO4-2
(aq)+2e−
