Chemistry - Online Test
Q1. Ethanoic
acid ----P/Br2 →2 – bromoethanoic acid. This reaction is called
Answer : Option C
Explaination / Solution:
No Explaination.
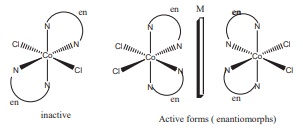
Q2. Which one of the following will give a pair of enantiomorphs?
Answer : Option B
Explaination / Solution:
Complexes given in other
options (a), (c) and (d) have symmetry elements and hence they are optically
inactive.

Q3. When aniline reacts with acetic anhydride the product formed is
Answer : Option D
Explaination / Solution:
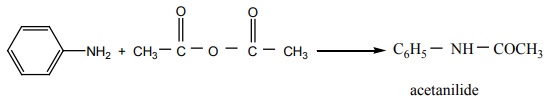
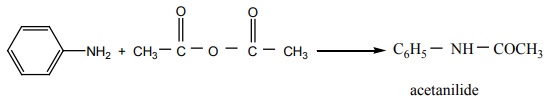
Q4. The composition of a sample
of wurtzite is Fe0.93 O1.00 what % of Iron present in the
form of Fe3+?
Answer : Option B
Explaination / Solution:
let
the number of Fe2+ ions
in the crystal be x
the number of Fe3+ ions
in the crystal be y
total number of Fe2+ and
Fe3+ ions is
x + y
given that x + y = 0.93
the total charge =0
x (2+) + (0.93 –x) (+ 3) −2 = 0
2x + 2.97 −3x −2 = 0
x = 0.79
Percentage of Fe3+
= [ ( 0.93 −0.79) / (0.93) ] 100 = 15.05%
Q5. In aqueous solution of amino acids mostly exists in,
Answer : Option D
Explaination / Solution:
No Explaination.
Q6. In a reversible reaction,
the enthalpy change and the activation energy in the forward direction are
respectively −x kJ mol−1 and y kJ mol−1 . Therefore , the energy of activation in the backward direction is
Answer : Option D
Explaination / Solution:
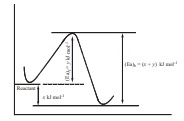
(x+y) kJmol-1
(x+y) 103 Jmol-1
Q7. Which one of the following is a bio-degradable polymer?
Answer : Option D
Explaination / Solution:
No Explaination.
Q8. The
percentage of pyridine (C5H5N) that forms pyridinium ion
(C5 H5NH) in a 0.10M aqueous pyridine solution (Kb
for C5 H5 N= 1.7 ×10-9
) is
Answer : Option B
Explaination / Solution:
C5H5N + H-OH ↔ C5H5 NH + OH-
(α2C) / (1-α ) =Kb
α2C =∼ Kb
α = √ [ Kb
/ C ] = √[ 1.7 ×10-9 / 0.1 ]
= √1.7 × 10-4
= √1.7 × 10−4 × 100
Percentage of dissociation = 1.3 ×10-2 = 0.013 %
Q9. Electrochemical process is used to extract
Answer : Option C
Explaination / Solution:
No Explaination.
Q10. The
number of electrons delivered at the cathode during electrolysis by a current
of 1A in 60 seconds is (charge of electron = 1.6 ×10−19 C )
Answer : Option C
Explaination / Solution:
Q =It
= 1A×60S
96500 C charge ≡ 6.022 ×1023
electrons
60 C charge ≡ [ 6.022 ×1023
/ 96500
] × 60
= 3.744 ×1020 electrons
