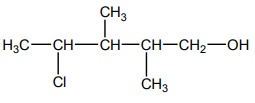
Chemistry - Online Test
Consider the following statements :
(i) increase in concentration of the reactant increases the rate
of a zero order reaction.
(ii) rate constant k is equal to collision frequency A if Ea
= 0
(iii) rate constant k is
equal to collision frequency A if Ea = °
(iv) a plot of ln(k) vs
T is a straight line
(v) a plot of ln (k) vs 1/T
is a straight line with a positive
slope.
In zero order reactions, increase in the concentration of reactant
does not alter the rate. So statement (i) is wrong.
k = A e – (Ea/RT)
if Ea = 0 so, statement
(ii) is correct, and statement (iii) is wrong
k = A e0
k = A
ln k = ln A – (Ea /R) (1/ T)
this equation is in the form of a straight line equatoion
y = c + m x
a plot of lnk vs 1/T is a straight line with negative slope
so statements (iv) and (v) are wrong.
HCOONa Basic in nature. + H ⋅OH ↔ NaOH strong base + H-COOH weak acid
C6H5NH3Cl-
+ H ⋅ OH ↔ H3O+Acidic +C6H5 -NH2
+ Cl-
KCN basic + H− OH ↔ KOH
strong base + HCN weak acid
basic, acidic, basic is correct.
m=ZIt
(mass of 1 mole of Cl2
gas = 71)
t = m/ ZI (∴mass of 0.1mole of Cl2 gas = 7.1 g mol−1 )
= 7.1 / [71/(2 ×96500) × 3 ]
(2 Cl- → Cl2 +2e- )
= (2 × 96500 × 7.1) / (71× 3)
= 6433.33sec
= 107.2 min
Assertion : bond dissociation energy of fluorine is greater than chlorine gas
Reason: chlorine has more electronic repulsion than fluorine