Chemistry - Online Test
Q1. The
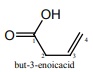
IUPAC name of 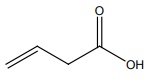

Answer : Option A
Explaination / Solution:

Q2. Which kind of isomerism is
possible for a complex [Co ( NH3)4 Br2] Cl
?
Answer : Option A
Explaination / Solution:
For [ MA4 B2 ]n+
complexes-geometrical isomerism is possible
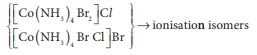
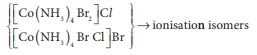
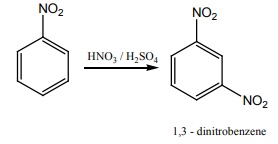
Q3. Nitrobenzene on reaction with Con HNO3 / H2SO4 at 80-100oC forms which one of the following products?
Answer : Option D
Explaination / Solution:

Q4. The vacant space in bcc lattice unit cell is
Answer : Option C
Explaination / Solution:
packing efficiency = 68%
therefore empty space
percentage = (100-68) = 32%
Q5. The pyrimidine bases present in DNA are
Answer : Option C
Explaination / Solution:
No Explaination.
Q6. In a first order reaction x → y ; if k is the rate constant and the initial concentration of the reactant x
is 0.1M, then, the half life is
Answer : Option C
Explaination / Solution:
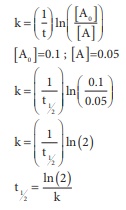
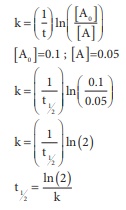
Q7. The polymer used in making blankets (artificial wool) is
Answer : Option A
Explaination / Solution:
No Explaination.
Q8. MY
and NY3 , are insoluble salts and have the same Ksp values
of 6.2 ×10-13 at room temperature.
Which statement would be true with regard to MY and NY3?
Answer : Option D
Explaination / Solution:
Addition of salt KY (having a
common ion Y–) decreases the solubility of MY and NY due to common ion effect.
Option (a) and (b) are wrong.
For salt MY , MY ↔ M+ + Y-
Ksp = (s)(s)
6.2 × 10-13 = s2
∴ s= √(6.2 × 10-13) = 10-7
for salt NY3 ,
NY3 ↔ N3+
+ 3Y-
Ksp = (s )(3s)3
Ksp = 27s4
s = [ (6.2 ×10−13)/27 ]1/4
s = 10-4
The molar solubility of MY in water is less
than of NY3
Q9. Zinc is obtained from ZnO by
Answer : Option A
Explaination / Solution:
No Explaination.
Q10. Zinc can be coated on iron to produce galvanized iron but the reverse is not possible. It is because
Answer : Option D
Explaination / Solution:
EoZn2+|Zn = − 0.76V and EoFe2+|Fe = −0.44V
Zinc has higher negative electrode potential than iron, iron cannot be coated
on zinc.
