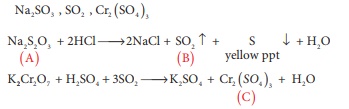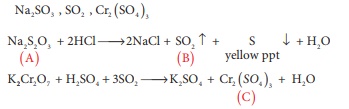Chemistry - Online Test
Q1. For a first order reaction,
the rate constant is 6.909 min-1.the time taken for 75% conversion
in minutes is
Answer : Option B
Explaination / Solution:
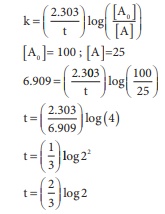
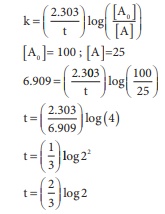
Q2. Which of the following is a co-polymer?
Answer : Option D
Explaination / Solution:
No Explaination.
Q3. If
the solubility product of lead iodide is 3.2 ×10-8
, its solubility will be
Answer : Option A
Explaination / Solution:
PbI2 (s) ↔ Pb2+
(aq) + 2I- (aq)
Ksp = (s)(2s)2
3.2 × 10-8 = 4s3
s =
(3.2×10-8 / 4)1/3
= (8 × 10-9 )1/3
= 2 × 10-3M
= 2 × 10-3M
Q4. In the extraction of aluminium from alumina by electrolysis, cryolite is added to
Answer : Option A
Explaination / Solution:
No Explaination.
Q5.
Among the following cells
I) Leclanche cell
II) Nickel – Cadmium cell
III) Lead storage battery
IV) Mercury cell
Answer : Option A
Explaination / Solution:
No Explaination.
Q6. Which of the following statements is not correct?
Answer : Option D
Explaination / Solution:
No Explaination.
Q7. Which one of the following is an example for homogeneous catalysis?
Answer : Option D
Explaination / Solution:
Both reactant and catalyst are in same phase. i.e(l)
Q8. Most easily liquefiable gas is
Answer : Option C
Explaination / Solution:
No Explaination.
Q9. Isopropylbenzene on air oxidation in the presence of dilute acid gives
Answer : Option D
Explaination / Solution:
phenol
Q10. A white crystalline salt
(A) react with dilute HCl to liberate a suffocating gas (B) and also forms a
yellow precipitate . The gas (B) turns potassium dichromate acidified with dil
H2SO4 to a green coloured solution(C). A,B and C are
respectively
Answer : Option A
Explaination / Solution: