Chemistry - Online Test
Q1. For a reaction Rate = k[acetone]3/2
acetone then unit of rate constant and rate of reaction respectively is
Answer : Option B
Explaination / Solution:


Q2. Nylon is an example of
Answer : Option A
Explaination / Solution:
No Explaination.
Q3. Which of the following fluro compounds is most likely to behave as a Lewis base?
Answer : Option B
Explaination / Solution:
BF3 → elctron deficient → Lewis
acid
PF3 → electron rich → lewis
base
CF4 → neutral → neither
lewis acid nor base
SiF4- → neutral → neither
lewis acid nor base
Q4. Match items in column - I
with the items of column – II and assign the correct code.
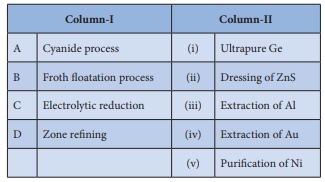

Answer : Option C
Explaination / Solution:
No Explaination.
Q5. How
many faradays of electricity are required for the following reaction to occur MnO-4
→ Mn2+
Answer : Option A
Explaination / Solution:
7MnO-4 + 5e− → Mn2+ + 4H2O
5 moles of electrons i.e., 5F
charge is required.
Q6. Oxidation state of carbon in its hydrides
Answer : Option A
Explaination / Solution:
Example : CH4+
in which the oxidation state of carbon is +4
Q7. Which one of the following is correctly matched?
Answer : Option B
Explaination / Solution:
No Explaination.
Q8. The basicity of
pyrophosphorous acid ( H4P2O5) is
Answer : Option B
Explaination / Solution:
No Explaination.
Q9. Which one of the following will react with phenol to give salicyladehyde after hydrolysis.
Answer : Option C
Explaination / Solution:
Riemer
– Tiemann reaction 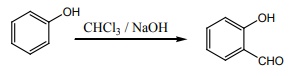

Q10. The correct order of increasing oxidizing power in the series
Answer : Option A
Explaination / Solution:
VO2+
{+5} < Cr2O72− {+6} < MnO4– {+7}
greater the oxidation state, higher is the xidizing power
