Chemistry - Online Test
Q1. The correct equation for the degree of an associating solute, 'n' molecules of which undergoes association in solution, is
Answer : Option C
Explaination / Solution:
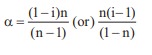
α=(1-i)n/(n-1) or n(i-1)/(1-n)
Q2. Shape
of ClF3 is
Answer : Option C
Explaination / Solution:
ClF3
- Sp3d hybridisation, 'T' shaped
Q3. In an organic compound, phosphorus is estimated as
Answer : Option A
Explaination / Solution:
No Explaination.
Q4. Which of the following is aliphatic saturated hydrocarbon
Answer : Option A
Explaination / Solution:
No Explaination.
Q5. Which of the following aqueous solutions has the highest boiling point ?
Answer : Option B
Explaination / Solution:
Elevation
of boiling point is more in the case of Na3PO4 (no. of
ions 4 ; 3 Na+, PO43–)
Q6. Non- Zero dipole moment is shown by
Answer : Option D
Explaination / Solution:

Q7. Ortho and para-nitro phenol can be separated by
Answer : Option C
Explaination / Solution:
No Explaination.
Q8. Identify the compound 'Z' in the following reaction
Answer : Option A
Explaination / Solution:
No Explaination.
Q9. The
freezing point depression constant for water is 1.86o K Kgmol-1.
If 5g Na2SO4 is dissolved in 45g water, the depression in
freezing point is 3.64oC. The Vant Hoff factor for Na2SO4
is
Answer : Option A
Explaination / Solution:
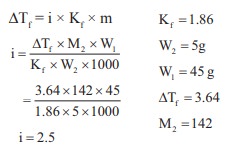
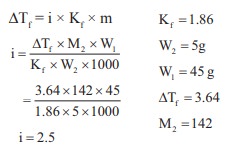
Q10. Which of the following conditions is not correct for resonating structures?
Answer : Option C
Explaination / Solution:
Correct statement is - the resonance hybrid should have lower energy than any of the contributing structure
