Chemistry - Online Test
Q1. The purity of an organic compound is determined by
Answer : Option D
Explaination / Solution:
No Explaination.
Q2. Peroxide effect (Kharasch effect) can be studied in case of
Answer : Option C
Explaination / Solution:
No Explaination.
Q3. Equimolal
aqueous solutions of NaCl and KCl are prepared. If the freezing point of NaCl
is –2ºC, the freezing point of KCl solution is expected to be
Answer : Option A
Explaination / Solution:
Equimolal aqueous solution of KCl also shows 2ºC depression in freezing point.
Q4. Among the following, the compound that contains, ionic, covalent and Co-ordinate linkage is
Answer : Option A
Explaination / Solution:
No Explaination.
Q5. A liquid which decomposes at its boiling point can be purified by
Answer : Option B
Explaination / Solution:
No Explaination.
Q6. 2 – butyne on chlorination gives
Answer : Option D
Explaination / Solution:
No Explaination.
Q7. Phenol dimerises in benzene having van't Hoff factor 0.54. What is the degree of association ?
Answer : Option D
Explaination / Solution:
i = 0.54
α=(1-i)n/(n-1)
=(1-0.54)2 / (2-1)
α= 0.92
Q8. CaO and NaCl have the same crystal structure and approximately the same radii. It U is the lattice energy of NaCl, the approximate lattice energy of CaO is
Answer : Option D
Explaination / Solution:
No Explaination.
Q9.
Assertion:
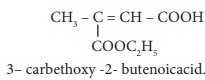 is
is
Answer : Option A
Explaination / Solution:
No Explaination.
Q10. Assertion
: An ideal solution obeys Raoults Law
Reason : In an ideal solution, solvent-solvent as well as
solute-solute interactions are similar to solute-solvent interactions.
Answer : Option A
Explaination / Solution:
both assertion and reason are correct and reason is the correct explanation of assertion.
