Chemistry - Online Test
At a given temperature and pressure
Volume α no. of moles
Volume α Mass / Molar mass
Volume α 28 / Molar mass
i.e. if molar mass is more, volume is less. Hence
HI has the least volume
A 20 litre container at 400 K contains CO2 (g) at
pressure 0.4 atm and an excess of SrO (neglect the volume of solid SrO). The
volume of the container is now decreased by moving the movable piston fitted in
the container. The maximum volume of the container, when pressure of CO2
attains its maximum value will be :
Given that : SrCO3 (S) ⇌ SrO (S) + CO2(g)
Given that KP = 1.6 atm
V1 = 20 L
V2 = ?
T1 = 400 K
T2 = 400 K
KP = PCO2
∴ PCO2=
1.6 atm
P1 = 0.4 atm. P2 = 1.6 atm
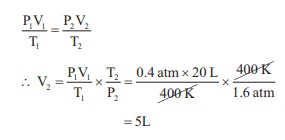
∆G = ∆H – T ∆ S
At 300K
∆G =30000 J mol–1 – 300 K x 100 J K–1
mol–1
∆G = 0
above 300 K ; ∆G will be negative and reaction
becomes spontaneous.
