Chemistry - Online Test
Q1. An organic Compound weighing 0.15g gave on carius estimation, 0.12g of silver bromide. The percentage of bromine in the Compound will be close to
Answer : Option B
Explaination / Solution:
No Explaination.
Q2. Which
of the following is/are true with respect to carbon -12.
Answer : Option A
Explaination / Solution:
(a)
relative atomic mass of C-12 is 12 u
Q3. Which of the following can be used as the halide component for friedal - crafts reaction ?
Answer : Option D
Explaination / Solution:
No Explaination.
Q4. Time
independent Schnodinger wave equation is
Answer : Option A
Explaination / Solution:
No Explaination.
Q5. 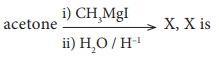
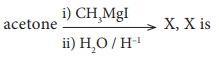
Answer : Option B
Explaination / Solution:
No Explaination.
Q6. When
CaC2 is heated in atmospheric nitrogen in an electric furnace the
compound formed is
Answer : Option B
Explaination / Solution:
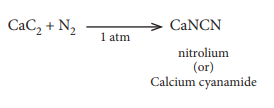
Q7. Which
of the following diagrams correctly describes the behaviour of a fixed mass of
an ideal gas ? (T is measured in K)
Answer : Option C
Explaination / Solution:
For a fixed mass of an ideal gas V α T P α 1/V
and PV = Constant
Q8.
The equilibrium constants of the following reactions are :
N2 + 3H2 ⇌ 2NH3 ; K1
N2 + O2 ⇌ 2NO ; K2
H2 + ½O2 ⇌ H2O ; K3
The equilibrium constant (K) for the reaction ;
2NH3 + 5/2 O2 ↔k↔ 2NO+3H2O+3H2O,
will be
Answer : Option C
Explaination / Solution:
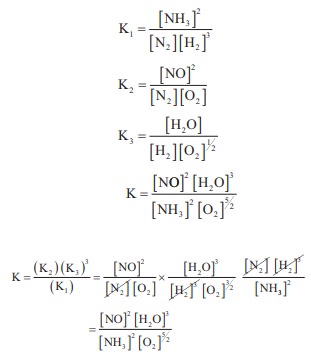
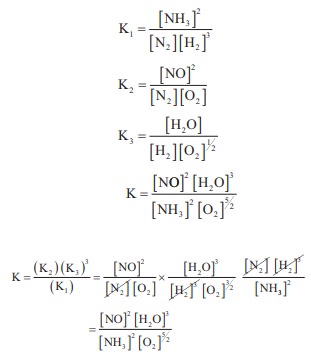
Q9. ∆S
is expected to be maximum for the reaction
Answer : Option D
Explaination / Solution:
In
CaCO3(S) → CaO(S) + CO2(g), entropy change is
positive. In (a) and (b) entropy change is negative ; in (c) entropy change is
zero.
Q10. What is the molality of a 10% W/W aqueous sodium hydroxide solution ?
Answer : Option B
Explaination / Solution:
10% W/W aqueous NaOH solution means that 10 g of sodium
hydroxide in 100g solution
Molality = no. of moles of solute / weight of solvent (in
kg)
= (10/40) / 0.1 =
0.25/0.1 = 2.5M
