Chemistry - Online Test
Q1. Some meta-directing substituents in aromatic substitution are given. Which one is most deactivating ?
Answer : Option B
Explaination / Solution:
No Explaination.
Q2. The
energy of an electron in the 3rd orbit of hydrogen atom is –E. The energy of an
electron in the first orbit will be
Answer : Option D
Explaination / Solution:
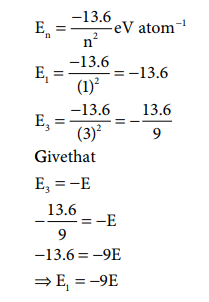
Q3. Chloroform reacts with nitric acid to produce
Answer : Option C
Explaination / Solution:
No Explaination.
Q4. Which
of the following pairs of elements exhibit diagonal relationship?
Answer : Option D
Explaination / Solution:
No Explaination.
Q5. Formula
of Gypsum is
Answer : Option A
Explaination / Solution:
CaSO4
. 2H2O∴
Option (a)
Q6. What
is the density of N2 gas at 227°C and 5.00 atm pressure? (R = 0.082
L atm K–1 mol–1)
Answer : Option C
Explaination / Solution:

Q7.
[Co(H2O)6]2+ (aq) (pink) +
4Cl– (aq) ⇌ [CoCl4]2–
(aq) (blue)+ 6 H2O (l)
Answer : Option A
Explaination / Solution:
on cooling, reverse reaction predominates and the solution
is pink in colour.
∴ decrease
in temperature, favours the reverse reaction ie reverse reaction is exothermic
and for the forward reaction is endothermic (ΔH > 0)
Q8. Molar
heat of vapourisation of a liquid is 4.8 kJ mol–1. If the entropy
change is 16 J mol–1 K–1, the boiling point of the liquid
is
Answer : Option B
Explaination / Solution:
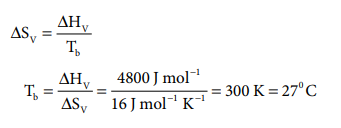
Q9. The Van't Hoff factor (i) for a dilute aqueous solution of the strong elecrolyte barium hydroxide is
Answer : Option D
Explaination / Solution:
Ba (OH)2 dissociates to form
Ba2+ and 2OH ion
α=(i-1) / (n-1)
i= α(n-1)+1
n=i=3 (for Ba(OH)2),
α =1)
Q10. Of the following molecules, which have shape similar to carbondixide?
Answer : Option C
Explaination / Solution:
Co2 - Linear
C2H2 - Linar
