Chemistry - Online Test
Q1. How
does electron affinity change when we move from left to right in a period in
the periodic table?
Answer : Option A
Explaination / Solution:
No Explaination.
Q2. The
name 'Blue John' is given to which of the following compounds ?
Answer : Option B
Explaination / Solution:
'Blue
john' – CaF2 (A variety of fluorite)
Q3. Assertion
: Critical temperature of CO2 is 304K, it can be liquefied above
304K.
Reason
: For a given mass of gas, volume is to directly proportional to pressure at constant
temperature
Answer : Option D
Explaination / Solution:
Correct
Statement: Critical temperature
of CO2 is 304 K. It means that CO2 cannot be liquefied
above 304 K, whatever the pressure may applied.
Pressure is inversely proportional to volume
Q4. Consider
the following reversible reaction at equilibrium, A + B ⇌ C, If the concentration of the reactants A and
B are doubled, then the equilibrium constant will
Answer : Option D
Explaination / Solution:
A + B ⇌ C
KC= [C] / [A] [B]
if [A] and [B] are doubled, [C] increases 4 times to
maintain KC as constant.
∴
equilibrium constant will remain the same – option (d)
Q5. In
an isothermal reversible compression of an ideal gas the sign of q, ∆S and w
are respectively
Answer : Option D
Explaination / Solution:
During compression, energy of the system increases,
in isothermal condition, to main-tain temperature constant, heat is liberated
from the system. Hence q is negative.
During compression entropy decreases.
During compression work is done on the system,
hence w is positive
Q6. 200ml
of an aqueous solution of a protein contains 1.26g of protein. At 300K, the
osmotic pressure of this solution is found to be 2.52 × 10–3 bar.
The molar mass of protein will be (R = 0.083 L bar mol–1K–1)
Answer : Option A
Explaination / Solution:
π = CRT
π = [ W/MV ] . RT
M = WRT / π V
= [1.26 x 0.083x300] / [2.52x10-3x0.2]
= 62.22 Kg mol-1
Q7. Xe
F2 is isostructural with
Answer : Option D
Explaination / Solution:
XeF2
is isostructural with ICl2–
Q8. The percentage of s-character of the hybrid orbitals in methane, ethane, ethene and ethyne are respectively
Answer : Option A
Explaination / Solution:
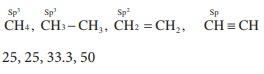
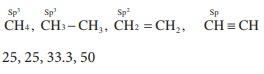
Q9. Sodium nitropruside reacts with sulphide ion to give a purple colour due to the for-mation of
Answer : Option C
Explaination / Solution:
No Explaination.
Q10. Which
of the following compound(s) has /have percentage of carbon same as that in
ethylene (C2H4)
Answer : Option A
Explaination / Solution:

