Chemistry - Online Test
Q1. The
units of Vander Waals constants 'b' and 'a' respectively
Answer : Option C
Explaination / Solution:
an2/V2 = atm
= atm L2/mol2 = L2
mol-2 atm
nb = L
b= L/mol = L mol-1
Q2.
Match the equilibria with the corresponding conditions,
i) Liquid ⇌ Vapour
ii) Solid ⇌ Liquid
iii) Solid ⇌ Vapour
iv) Solute (s) ⇌ Solute
(Solution)
1. melting point
2. Saturated solution
3. Boiling point
4. Sublimation point
5. Unsaturated solution
Answer : Option B
Explaination / Solution:
No Explaination.
Q3. The
temperature of the system, decreases in an ___________________
Answer : Option C
Explaination / Solution:
No Explaination.
Q4. For
a solution, the plot of osmotic pressure (p) verses the concentration (c in mol
L–1) gives a straight line with slope 310R where 'R' is the gas
constant. The temperature at which osmotic pressure measured is
Answer : Option C
Explaination / Solution:
π = CRT
y = x (m)
m = RT
310 R = RT
∴ T = 310 K
= 37º C
Q5. The types of hybridiration on the five carbon atom from right to left in the, 2,3 pentadiene.
Answer : Option A
Explaination / Solution:
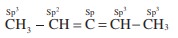
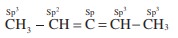
Q6. Connect pair of compounds which give blue colouration / precipitate and white precipi-tate respectively, when their Lassaigne’s test is separately done.
Answer : Option D
Explaination / Solution:
No Explaination.
Q7. Which
of the following contain same number of carbon atoms as in 6 g of carbon-12.
Answer : Option C
Explaination / Solution:
No. of moles of carbon present in 6 g of C-12 =
Mass / Molar mass
=6/12 = 0.5 moles = 0.5 × 6.022 × 1023
carbon atoms. No. of moles in 8 g of methane = 8 / 16 = 0.5 moles
=0.5 × 6.022 × 1023 carbon atoms.
=2 × 0.25 × 6.022 × 1023 carbon atoms.
Q8. Which of the following compounds will not undergo Friedal – crafts reaction easily ?
Answer : Option A
Explaination / Solution:
No Explaination.
Q9. The
ratio of de Broglie wavelengths of a deuterium atom to that of an α - particle,
when the velocity of the former is five times greater than that of later, is
Answer : Option D
Explaination / Solution:
No Explaination.
Q10. The raw material for Rasching process
Answer : Option C
Explaination / Solution:
No Explaination.
