Chemistry - Online Test
Q1. The
mass of a non-voltaile solute (molar mass 80 g mol–1) which should
be dissolved in 92g of toluene to reduce its vapour pressure to 90%
Answer : Option D
Explaination / Solution:
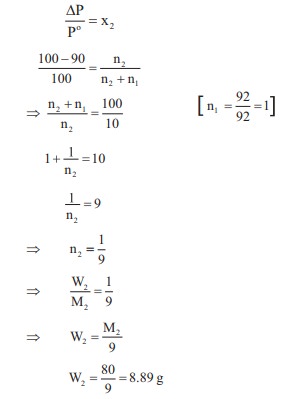
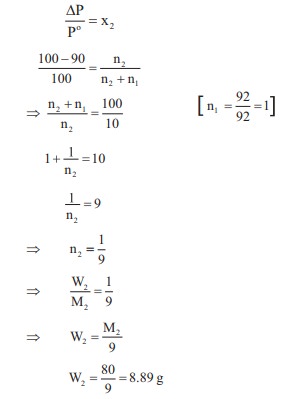
Q2. Some
of the following properties of two species, NO3- and H3O+
are described below. which one of them is correct?
Answer : Option A
Explaination / Solution:
NO3– - Sp2 hybridisation,
planar
H3O+ - Sp3 hybridisation,
pyramidal
Q3. Lassaigne’s test for the detection of nitrogen fails in
Answer : Option C
Explaination / Solution:
No Explaination.
Q4. The
mass of a gas that occupies a volume of 612.5 ml at room temperature and
pressure (250 c and 1 atm pressure) is 1.1g. The molar mass of the
gas is
Answer : Option B
Explaination / Solution:
No. of moles of a gas that occupies a volume of
612.5 mL at room temperature and pressure (25 0 C and 1 atm
pressure)
=612.5 × 10-3 L / 24.5 Lmol-1
=0.025 moles
We know that,
Molar mass
=Mass / no. of moles
=1.1 g
/0.025 mol = 44 g mol-1
Q5. 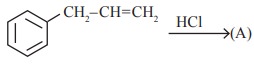 is
is
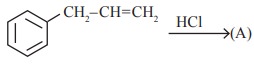 is
is
Answer : Option D
Explaination / Solution:
No Explaination.
Q6. A
macroscopic particle of mass 100 g and moving at a velocity of 100 cm s–1
will have a de Broglie wavelength of
Answer : Option C
Explaination / Solution:
m = 100 g = 100 x 10-3 kg
= 100 cm s-1 = 100 x 10-2 m s-1
λ =h/mv
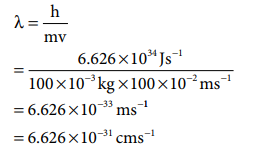
Q7. Ethylidene chloride on treatment with aqueous KOH gives
Answer : Option A
Explaination / Solution:
No Explaination.
Q8. Which
one of the following is true about metallic character when we move from left to
right in a period and top to bottom in a group?
Answer : Option A
Explaination / Solution:
No Explaination.
Q9. Water
is a
Answer : Option C
Explaination / Solution:
Water
is a amphoteric oxide.
Q10. Which
of the following statement is false ?
Answer : Option A
Explaination / Solution:
Ca2+
ion plays an important role in maintaining regular heart beat.
