Chemistry - Online Test
Q1. What
is the mass of precipitate formed when 50 ml of 8.5 % solution of AgNO3
is mixed with 100 ml of 1.865 % potassium chloride solution?
Answer : Option A
Explaination / Solution:
AgNO3 + KCl → KNO3 + AgCl
50 mL of 8.5 % solution contains 4.25 g of AgNO3
No. of moles of AgNO3 present in 50 mL of 8.5 %
AgNO3 solution
=Mass / Molar mass
=4.25 / 170
=0.025 moles
Similarly, No of moles of KCl present in 100 mL of
1.865 % KCl solution
=1.865 / 74.5
=0.025 moles
So total amount of AgCl formed is 0.025 moles
(based on the stoichiometry)
Amount of
AgCl present in 0.025 moles of AgCl
=no. of moles x molar mass
=0.025 x
143.5 = 3.59 g
Q2. Propyne on passing through red hot iron tube gives
Answer : Option A
Explaination / Solution:
No Explaination.
Q3. If
uncertainty in position and momentum are equal, then minimum uncertainty in
velocity is
Answer : Option C
Explaination / Solution:
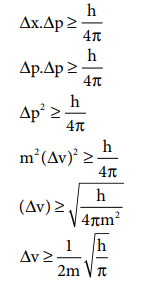
Q4. Which one of the following is most reactive towards nucleophilic substitution reaction ?
Answer : Option D
Explaination / Solution:
No Explaination.
Q5. The
First ionisation potential of Na, Mg and Si are 496, 737 and 786 kJ mol-1
respectively. The ionisation potential of Al will be closer to
Answer : Option B
Explaination / Solution:
No Explaination.
Q6.
Heavy
water is used as
Answer : Option C
Explaination / Solution:
Heavy
water is used as moderator as well as coolant in nuclear reactions.
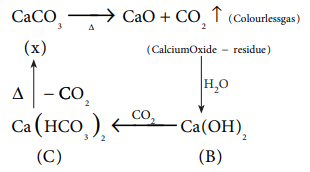
Q7. The
compound (X) on heating gives a colourless gas and a residue that is dissolved
in water to obtain (B). Excess of CO2 is bubbled through aqueous
solution of B, C is formed. Solid (C) on heating gives back X. (B) is
Answer : Option B
Explaination / Solution:
Q8. Maximum
deviation from ideal gas is expected from
Answer : Option B
Explaination / Solution:
No Explaination.
Q9. For
the formation of Two moles of SO3(g) from SO2 and O2,
the equilibrium constant is K1. The equilibrium constant for the
dissociation of one mole of SO3 into SO2 and O2
is
Answer : Option C
Explaination / Solution:


Q10. The
correct thermodynamic conditions for the spontaneous reaction at all
temperature is
Answer : Option A
Explaination / Solution:
No Explaination.
