Chemistry - Online Test
Q1. The
major products obtained when chlorobenzene is nitrated with HNO3 and
con H2SO4
Answer : Option A
Explaination / Solution:
No Explaination.
Q2. Which
of the following orders of ionic radii is correct?
Answer : Option D
Explaination / Solution:
No Explaination.
Q3. The
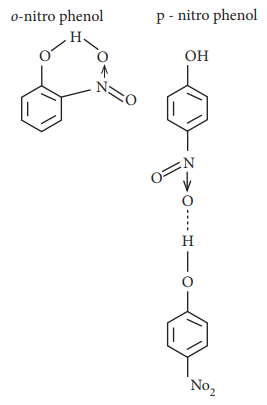
type of H-bonding present in ortho nitro phenol and p-nitro phenol are respectively
Answer : Option C
Explaination / Solution:

Q4. A
colourless solid substance (A) on heating evolved CO2 and also gave
a white resi-due, soluble in water. Residue also gave CO2 when
treated with dilute HCl.
Answer : Option B
Explaination / Solution:
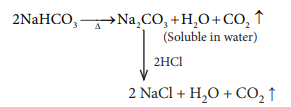
Q5. Four
gases P, Q, R and S have almost same values of 'b' but their 'a' values (a, b
are Vander Waals Constants) are in the order Q < R < S < P. At a
particular temperature, among the four gases the most easily liquefiable one is
Answer : Option A
Explaination / Solution:
Greater
the 'a' value, easier the liquefaction
Q6. Which of the following is not a general characteristic of equilibrium involving physical process
Answer : Option C
Explaination / Solution:
correct statement : Physical processes occurs at the same
rate at equilibrium
∴ option
(c) is incorrect statement
Q7. The
bond dissociation energy of methane and ethane are 360 kJ mol–1 and
620 kJ mol–1 respec-tively. Then, the bond dissociation energy of
C-C bond is
Answer : Option D
Explaination / Solution:
4EC–H= 360 kJ mol–1
EC–H= 90 kJ mol–1
EC–C + 6 EC–H = 620 kJ mol–1
EC–C + 6 × 90 = 620 kJ mol–1
EC–C + 540= 620 kJ mol–1
EC–C= 80 kJ mol–1
Q8. The
relative lowering of vapour pressure of a sugar solution in water is 3.5 × 10–3.
The mole fraction of water in that solution is
Answer : Option D
Explaination / Solution:
∆P / Pº =
Xsugar
3.5 × 10–3 = Xsugar
Xsugar + XH2o = 1
XH2o = 1 –
0.0035 = 0.9965
Q9. In
which of the following molecules / ions BF3, NO2-,
H2 O the central atom is sp2 hybridised?
Answer : Option C
Explaination / Solution:
H2O - Central atom Sp3 hybridised
NO2– - Central atom Sp2 hybridised
BF3 - Central atom Sp2 hybridised
NH2– - Central atom Sp3 hybridised
option
Q10. Nitrogen detection in an organic compound is carried out by Lassaigne’s test. The blue colour formed is due to the formation of.
Answer : Option B
Explaination / Solution:
No Explaination.
