Chemistry - Online Test
Q1. The
suspension of slaked lime in water is known as
Answer : Option C
Explaination / Solution:
Slaked lime Ca(OH)2
The suspension is called milk of lime and the clear
solution is called lime water.
Q2. The
variation of volume V, with temperature T, keeping pressure constant is called
the coefficient of thermal expansion ie α = 1/V (∂V/∂T)P. For an
ideal gas α is equal to
Answer : Option B
Explaination / Solution:
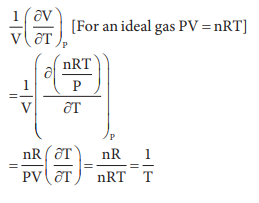
Q3. In
a chemical equilibrium, the rate constant for the forward reaction is 2.5 × 102
and the equilibrium constant is 50. The rate constant for the reverse reaction
is,
Answer : Option B
Explaination / Solution:
Kf = 2.5 × 102
KC = 50
Kr = ?
Kc = Kf/Kr
50 = 2.5 x 102/ Kr
Kr = 5
Q4. When
15.68 litres of a gas mixture of methane and propane are fully combusted at 00
C and 1 atmosphere, 32 litres of oxygen at the same temperature and pressure
are consumed. The amount of heat of released from this combustion in kJ is (∆HC
(CH4) = – 890 kJ mol–1 and ∆HC (C3H8)
= – 2220 kJ mol–1)
Answer : Option D
Explaination / Solution:
Given :
ΔHC (CH4)= – 890 kJ
mol–1
ΔHC (C3H8)=
– 2220 kJ mol–1

Q5. Two liquids X and Y on mixing gives a warm solution. The solution is
Answer : Option D
Explaination / Solution:
ΔHmix
is negative and show negative deviation from Raoults law.
Q6. The molecules having same hybridisation, shape and number of lone pairs of electons are
Answer : Option A
Explaination / Solution:
SeF4, XeO2F2 - Sp3d
hybridisation,
First option T-shaped, one line pair on central atom.
Q7. 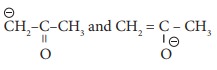 are
are
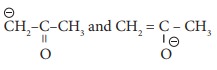 are
are
Answer : Option B
Explaination / Solution:
No Explaination.
Q8. Two
22.4 litre containers A and B contains 8 g of O2 and 8 g of SO2
respectively at 273 K and 1 atm pressure, then
Answer : Option C
Explaination / Solution:
No, of moles of oxygen=8 g/32 g
=0.25 moles of oxygen
No. of moles of sulphur dioxide=8 g / 64 g
=0.125 moles of sulphur dioxide
Ratio between the no. of molecules=0.25: 0.125
=2:1
Q9. In which of the following molecules, all atoms are co-planar
Answer : Option D
Explaination / Solution:
No Explaination.
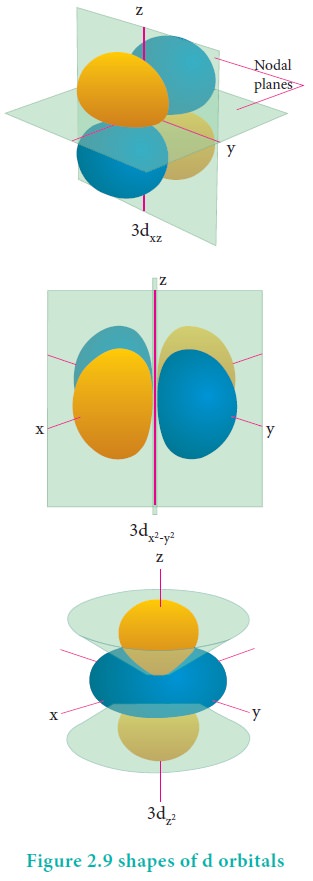
Q10. Electron
density in the yz plane of 3dx 2 −y2
orbital is
Answer : Option A
Explaination / Solution:
Option
(a) - Zero (Refer to Figure 2.9)