Chemistry - Online Test
Q1. Given
that C(g) + O2 (g) → CO2 (g) ∆H0 = – a kJ; 2
CO(g) + O2(g) → 2CO2(g) ∆H0 = –b kJ; Calculate
the ∆H0 for the reaction C(g) + ½O2(g) → CO(g)
Answer : Option D
Explaination / Solution:
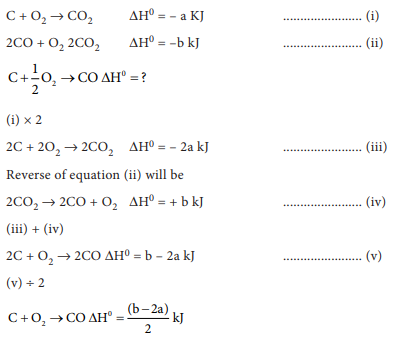
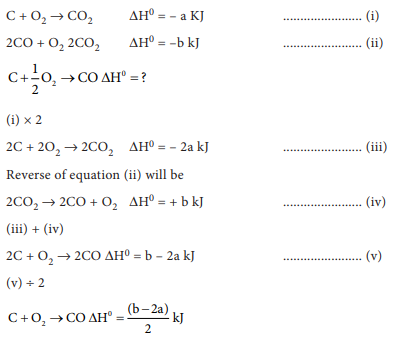
Q2. Normality of 1.25M sulphuric acid is
Answer : Option C
Explaination / Solution:
Normality of H2SO4 = (no.of replacable
H+)×M
= 2 × 1.25
= 2.5 N
Q3. Pick out the incorrect statement from the following
Answer : Option C
Explaination / Solution:
Correct statement : All five Sp3d hybrid orbitals
are equivalent
incorrect statement : Option (c)
Q4. Which one of the following shows functional isomerism?
Answer : Option C
Explaination / Solution:
No Explaination.
Q5. If
Avogadro number were changed from 6.022 × 1023 to 6.022 x 1020, this would change
Answer : Option D
Explaination / Solution:
The
mass of one mole of carbon
Q6. Consider
the nitration of benzene using mixed con H2SO4 and HNO3
if a large quantity of KHSO4 is added to the mixture, the rate of
nitration will be
Answer : Option D
Explaination / Solution:
No Explaination.
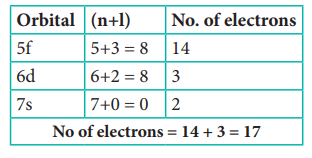
Q7. How
many electrons in an atom with atomic number 105 can have (n + l) = 8 ?
Answer : Option B
Explaination / Solution:
n+l = 8
Electronic configuration of atom with atomic number
105 is [Rn] 5f14 6d3 7s2
Q8. The carbo cation formed in SN1 reaction of al kyl halide in the slow step is
Answer : Option B
Explaination / Solution:
No Explaination.
Q9. In
a given shell the order of screening effect is
Answer : Option A
Explaination / Solution:
No Explaination.
Q10. In
solid ice, oxygen atom is surrounded
Answer : Option A
Explaination / Solution:
(a)
tetrahedrally surrounded by 4
hydrogen atoms (refer
4.6 (a) Structure of ice)
