Chemistry - Online Test
Q1. The
equivalent mass of ferrous oxalate is
Answer : Option C
Explaination / Solution:
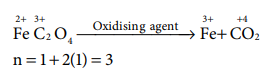
Q2. 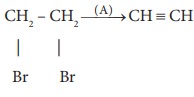 ,where A is,
,where A is,
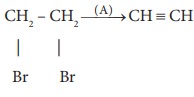 ,where A is,
,where A is,
Answer : Option C
Explaination / Solution:
No Explaination.
Q3. Consider
the following sets of quantum numbers :

Which
of the following sets of quantum number is not possible ?
Answer : Option B
Explaination / Solution:
(ii) l
can have the values from 0 to n-1
n=2; possible 'l' values are 0, 1 hence l = 2 is not possible.
(iv)for l=0;
m=-1 not possible
(v)for n=3 l
= 4 and m = 3 not possible
Q4. The most easily hydrolysed molecule under SN1 condition is
Answer : Option A
Explaination / Solution:
No Explaination.
Q5. IE1
and IE2 of Mg are 179 and 348 kcal mol-1 respectively.
The energy required for the reaction Mg → Mg2+ + 2 e- is
Answer : Option C
Explaination / Solution:
No Explaination.
Q6.
Assertion
(A): Oxygen plays a key role
in the troposphere
![]()
![]() Reason
(R): Troposphere is not responsible
for all biological activities
Reason
(R): Troposphere is not responsible
for all biological activities
Answer : Option D
Explaination / Solution:
No Explaination.
Q7. The
reaction H3PO2 + D2O → H2DPO2
+ HDO indicates that hypo-phosphorus acid is
Answer : Option C
Explaination / Solution:
Hypophosphorus acid on reaction with D2O,
only one hydrogen is replaced by deuterium and hence it is mono basic
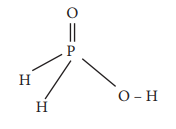
Option (c) monobasic acid
Q8. In
context with beryllium, which one of the following statements is incorrect ?
Answer : Option C
Explaination / Solution:
Correct Statement: Beryllium salts are easily hydrolysed
Q9. Equal
moles of hydrogen and oxygen gases are placed in a container, with a pin-hole
through which both can escape what fraction of oxygen escapes in the time
required for one-half of the hydrogen to escape.
Answer : Option C
Explaination / Solution:
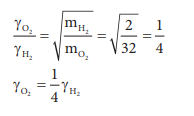
The fraction of oxygen that escapes in the time
required for one half of the hydrogen to escape is 1/8
Q10. Equimolar
concentrations of H2 and I2 are heated to equilibrium in
a 1 litre flask. What percentage of initial concentration of H2 has
reacted at equilibrium if rate constant for both forward and reverse reactions
are equal
Answer : Option A
Explaination / Solution:
V = 1L
H2 + I2 ⇌ 2HI
[H2]initial = [I2]initial
= a
[H2]eq = [I2]eq
= (a – x)
and [HI]eq = 2x
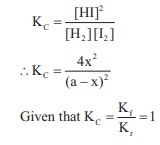
∴ 4x2
= (a – x)2
4x2 = a2 + x2 – 2ax
3x2 + 2ax – a2 = 0
x = –a & x = a/3
degree of dessociation = a/3× 100
= 33.33 %
