Chemistry - Online Test
Q1. Which
of the following is second most electronegative element?
Answer : Option A
Explaination / Solution:
No Explaination.
Q2.
Assertion
(A): Excessive use of chlorinated
pesticide causes soil and water pollution.
Reason (R)
: Such pesticides are non-biodegradable.
Answer : Option A
Explaination / Solution:
No Explaination.
Q3. The
hybridisation of oxygen atom is H2O and H2O2
are, respectively
Answer : Option D
Explaination / Solution:
No Explaination.
Q4. Which
is the correct sequence of solubility of carbonates of alkaline earth metals ?
Answer : Option B
Explaination / Solution:
Solubility of carbonates decreases down the group
and hence the correct order of solubility is,
MgCO3 > CaCO3 > SrCO3
> BaCO3
Q5. At
identical temperature and pressure, the rate of diffusion of hydrogen gas is
3√3 times that of a hydrocarbon having molecular formula CnH2n–2.
What is the value of n ?
Answer : Option B
Explaination / Solution:

Q6.
Consider the reaction where KP = 0.5 at a
particular temperature
PCl5(g) ⇌ PCl3
(g) + Cl2 (g)
if the three gases are mixed in a container so that the
partial pressure of each gas is initially 1 atm, then which one of the
following is true
Answer : Option C
Explaination / Solution:
KP = 0.5
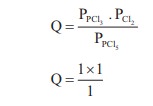
Q > KP ∴ Reverse
reaction is favoured ; i.e. more PCl5 will be produced. option (c)
Q7. The
value of ∆H for cooling 2 moles of an ideal monatomic gas from 125° C to 25° C
at constant pressure will be given [CP=5/2 R]
Answer : Option B
Explaination / Solution:
Ti = 125° C = 398 K
Tf = 25° C = 298 K
ΔH = nCp (Tf – Ti)
ΔH = 2x(5/2)R(298-398)
ΔH = – 500 R
Q8. The
KH for the solution of oxygen dissolved in water is 4 × 104
atm at a given temperature. If the partial pressure of oxygen in air is 0.4
atm, the mole fraction of oxygen in solution is
Answer : Option C
Explaination / Solution:


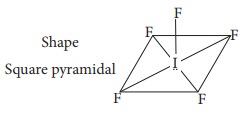
Q9. Shape
and hybridisation of IF5 are
Answer : Option C
Explaination / Solution:
IF5
- 5 bond pair + 1 lone pair hybridisation Sp3d2

Q10. How
many cyclic and acyclic isomers are possible for the molecular formula C3H6O?
Answer : Option C
Explaination / Solution:
No Explaination.
