Chemistry - Online Test
Q1. If
temperature and volume of an ideal gas is increased to twice its values, the
initial pressure P becomes
Answer : Option C
Explaination / Solution:
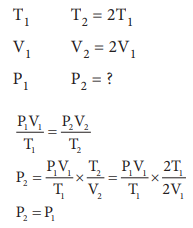
Q2.
In the reaction,
Fe (OH)3 (s) ⇌ Fe3+(aq)
+ 3OH–(aq),
if the concentration of OH– ions is decreased by
¼ times, then the equilibrium concentration of Fe3+ will
Answer : Option D
Explaination / Solution:
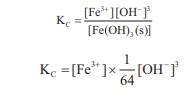
To
maintain KC as constant, concentration of Fe3+ will
increase by 64 times.
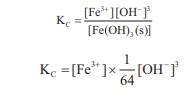
Q3. The
work done by the liberated gas when 55.85 g of iron (molar mass 55.85 g mol–1)
reacts with hydrochloric acid in an open beaker at 25° C
Answer : Option A
Explaination / Solution:
Fe + 2HCl → FeCl2 + H2
1 mole of
Iron liberates 1 mole of
Hydrogen gas
55.85 g Iron
= 1 mole Iron
∴ n = 1
T = 25° C =
298 K
w = – P Δ V
w =-P (nRT/P)
w = – nRT
w = –1 ×
8.314 × 298 J
w = –
2477.57 J
w = – 2.48
kJ
Q4. The
empirical formula of a non-electrolyte(X) is CH2O. A solution
containing six gram of X exerts the same osmotic pressure as that of 0.025M
glucose solution at the same temperature. The molecular formula of X is
Answer : Option B
Explaination / Solution:
(π1)non electrolyte = (π2)glucose
C1RT = C2RT
[CH2O
=> 12+2+16=30]
W1/M1 = W1/M2
6 / n(30) = 0.025
n = 8
∴ molecular
formula = C8H16O8
Q5. Bond order of a species is 2.5 and the number of electons in its bonding molecular orbital is formd to be 8 The no. of electons in its antibonding molecular orbital is
Answer : Option A
Explaination / Solution:
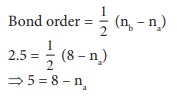
5 = 8 – na
na = 8-5 = 3
Q6. The isomer of ethanol is
Answer : Option B
Explaination / Solution:
No Explaination.
Q7. The
correct increasing order of the oxidation state of sulphur in the anions SO42-,
SO32- , S2O42-,
S2O62- is
Answer : Option C
Explaination / Solution:
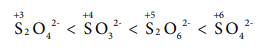
Q8. Identify
the compound (A) in the following reaction
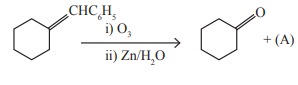
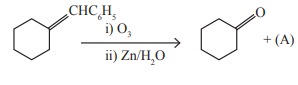
Answer : Option A
Explaination / Solution:
No Explaination.
Q9. If
n = 6, the correct sequence for filling of electrons will be,
Answer : Option A
Explaination / Solution:
n = 6
According Aufbau principle,
6s →4f →5d →6p
ns → (n-1)f →(n-2)d → np
Q10. Freon-12 is manufactured from tetrachloro methane by
Answer : Option B
Explaination / Solution:
No Explaination.
