Chemistry - Online Test
Q1.
Assertion:
In
mono haloarenes, electrophilic
substitution occurs at ortho and para positions.
Reason: Halogen
atom is a ring deactivator
Answer : Option B
Explaination / Solution:
No Explaination.
Q2.
Assertion: Helium has the highest value of
ionisation energy among all the elements known
Answer : Option C
Explaination / Solution:
No Explaination.
Q3. 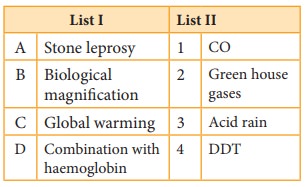

Answer : Option B
Explaination / Solution:
No Explaination.
Q4. For
decolourisation of 1 mole of acidified KMnO4, the moles of H2O2
required is
Answer : Option C
Explaination / Solution:
2MnO4–
+ 5H2O2(aq) + 6H+ → 2Mn2+ + 5O2
+ 8H2O
Q5. Assertion
: Generally alkali and alkaline earth metals form superoxides
Reason
: There is a single bond between O and O in superoxides.
Answer : Option D
Explaination / Solution:
Among alkali and alkaline earth metals,
K, Rb and Cs alone forms superoxides.
Superoxide O2– has 3 electron
bond.
Q6. Compressibility
factor for CO2 at 400 K and 71.0 bar is 0.8697. The molar volume of
CO2 under these conditions is
Answer : Option C
Explaination / Solution:

Q7.
The values of K P1 and KP2 for the reactions
X ⇌ Y + Z
A ⇌ 2B are in
the ratio 9 : 1 if degree of dissociation and initial concentration of X and A
be equal then total pressure at equilibrium P1, and P2
are in the ratio
Answer : Option A
Explaination / Solution:


Q8. Change
in internal energy, when 4 kJ of work is done on the system and 1 kJ of heat is
given out by the system is
Answer : Option C
Explaination / Solution:
ΔU = q + w
ΔU = – 1 kJ + 4 kJ
ΔU = + 3kJ
Q9. At same temperature, which pair of the following solutions are isotonic ?
Answer : Option D
Explaination / Solution:
0.1 × 3 ion [Ba2+,
2NO3–]
= 0.1 × 3 ion [2 Na+,
SO4–]
Q10. Which one of the following is diamagnetic.?
Answer : Option B
Explaination / Solution:
O22–
is diamagnetic. Additional two electrons paired in antibonding molecular
orib-ital π*2Py and π*2 Pz
