Chemistry - Online Test
i. Atmospheric pressure is less at the top of a
mountain than at sea level
ii. Gases are much more compressible than solids or
liquids
iii. When the atmospheric pressure increases the
height of the mercury column rises Select the correct statement
If x is the fraction of PCl5 dissociated at
equilibrium in the reaction
PCl5 ⇌ PCl3
+ Cl2
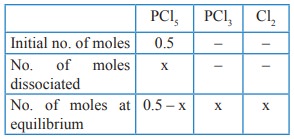
Total no. of moles at equilibrium = 0.5–x + x + x
O2 > O3 > H2O2
2 > 1.5 > 1
7.5 g of gas occupies a volume of 5.6 liters at 273
K and 1 atm pressure Therefore,
the mass of gas that occupies a volume of 22.4 liters
=7.5g / 5.6 L = 30g
Molar mass of NO (14+16) =
30 g
Assertion : Number of radial and angular nodes for
3p orbital are 1, 1 respectively.
No. of radial node = n-l-1
No. of angular node = l
for 3p orbital
No. of angular node = l = 1
No. of radial node = n-l-1 = 3-1-1 = 1
