Chemistry - Online Test
No. of moles of water present in 180 g
= Mass of water / Molar mass of water
= 180 g / 18 g mol-1 = 10 moles
One mole of water contains
= 6.022 x 1023 water molecules
10 mole of water contains = 6.022 x 1023
x 10 = 6.022 x 1024 water molecules
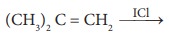
n = 3; l=1; m = -1
either 3px or 3py
i.e., Maximum two electrons can be accomodated
either in 3px or in 3py
Match the compounds given in Column I with suitable items
given in Column II
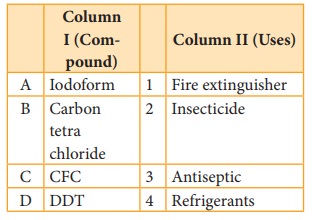
Code
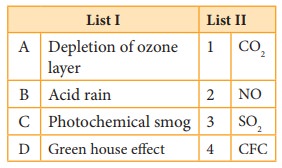
Match the flame colours of the alkali and alkaline
earth metal salts in the bunsen burner
(p) Sodium (1) Brick red
(q) Calcium (2) Yellow
(r) Barium (3) Violet
(s) Strontium (4) Apple green
(t) Cesium (5) Crimson red
(u) Potassium (6) Blue
(p) sodium –yellow (2)
(q) Calcium – Brick red (1)
(r) Barium – apple green (4)
(s) Strontium – Crimson red (5)
(t) Cesium – blue (6)
(u) Potassium – Violet (3)
