Chemistry - Online Test
Q1. Which of the following reagent is helpful to differentiate ethylene dichloride and ethylidene chloride?
Answer : Option C
Explaination / Solution:
No Explaination.
Q2. The
element with positive electron gain enthalpy is
Answer : Option C
Explaination / Solution:
No Explaination.
Q3. Biochemical oxygen Demand value less than 5 ppm indicates a water sample to be
Answer : Option C
Explaination / Solution:
No Explaination.
Q4. A
commercial sample of hydrogen peroxide marked as 100 volume H2O2,
it means that
Answer : Option A
Explaination / Solution:
(a)
1 mL of H2O2 will give 100ml O2 at STP.
Q5. Which
of the following has highest hydration energy
Answer : Option A
Explaination / Solution:
MgCl2
The order of hydration energy of alkaline earth
metal is
Be2+ > Mg2+ > Ca2+
> Sr2+ > Ba2+
Q6.
The gas which can be most easily liquefied is
The table indicates the value of van der Waals
constant ‘a’ in (dm3)2atm. mol-2
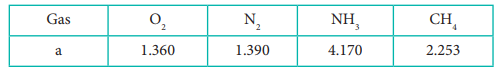
Answer : Option C
Explaination / Solution:
Higher
the value of 'a', greater the intermolecular force of attraction, easier the liquefaction.
Q7. In
which of the following equilibrium, KP and KC are not
equal?
Answer : Option D
Explaination / Solution:
For reaction given in option (a), (b) & (c) Δng = 0
For option (d) Δng = 2 – 1 = 1
∴ KP
= KC (RT)
Q8. Which
of the following is not a thermodynamic function ?
Answer : Option D
Explaination / Solution:
No Explaination.
Q9. At
100oC the vapour pressure of a solution containing 6.5g a solute in
100g water is 732mm. If Kb = 0.52, the boiling point of this solution
will be
Answer : Option C
Explaination / Solution:

Tb – 100 = 1.06
Tb = 100 + 1.06
= 101.06 ≈ 101o C
Q10. Hybridisation
of central atom in PCl5 involves the mixing of orbitals.
Answer : Option C
Explaination / Solution:
PCl5 – Sp3d hybridization
S, Px , Py , Pz and dx2 −y2
