Chemistry - Online Test
Q1. Use
of hot air balloon in sports at meteorological observation is an application of
Answer : Option A
Explaination / Solution:
No Explaination.
Q2. For
the reaction AB (g) ⇌ A(g) + B(g), at equilibrium, AB is 20% dissociated at
a total pressure of P, The equilibrium constant KP is related to the
total pressure by the expression
Answer : Option A
Explaination / Solution:
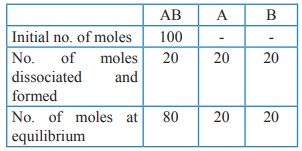
Total no. of moles at equilibrium = 80 + 20 + 20 = 120

Q3. The
enthalpies of formation of Al2O3 and Cr2O3
are – 1596 kJ and – 1134 kJ, respectively. ∆H for the reaction 2Al + Cr2O3
→ 2Cr + Al2O3 is
Answer : Option D
Explaination / Solution:
2Al + Cr2O3 → 2Cr + Al2O3
ΔHr0 = [2ΔHf (Cr) + ΔHf (Al2O3)]–
[2ΔHf (Al) + ΔHf
(Cr2O3)] ΔHr0 = [0 + (–
1596 kJ)]– [0 + (– 1134)]
ΔHr0 = – 1596
kJ + 1134 kJ
ΔHr0 = – 462
kJ
Q4. The Henry's law constants for two gases A and B are x and y respectively. The ratio of mole fractions of A to B is 0.2. The ratio of mole fraction of B and A dissolved in water will be
Answer : Option D
Explaination / Solution:
Given, (KH)A = x
(KH)B = y
xA/xB = 0.2
(xA/xB)in solution = ?
PA = x (xA) in solution –
(1)
PB = y (xB) in solution –
(2)
(xA/xB)in solution = (PB/PA)
. (x/y) = (xB/xA) . (x/y) = (1/0.2) . (x/y) = 5x/y
Q5. Which of these represents the correct order of their increasing bond order.
Answer : Option D
Explaination / Solution:
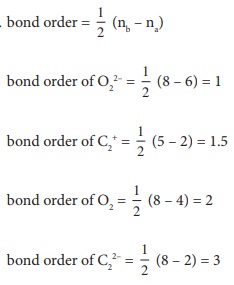
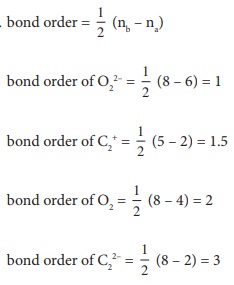
Q6. The
IUPAC name of 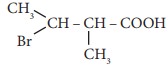 is
is
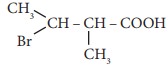 is
is
Answer : Option C
Explaination / Solution:
No Explaination.
Q7. Heterolytic fission of C-Br bond results in the formation of
Answer : Option D
Explaination / Solution:
No Explaination.
Q8. The
equivalent mass of potassium permanganate in alkaline medium is MnO4-
+ 2H2O+3e- →
MnO2 + 4OH-
Answer : Option B
Explaination / Solution:
The reduction reaction of the oxidising agent(MnO4–)
involves gain of 3 electrons.
Hence the equivalent mass = (Molar mass of KMnO4)/3
= 158.1/3 = 52.7
Q9. The major product formed when 2 – bromo – 2 – methyl butane is refluxed with ethanolic KOH is
Answer : Option A
Explaination / Solution:
No Explaination.
Q10. For
d-electron, the orbital angular momentum is
Answer : Option D
Explaination / Solution:
Orbital angular momentum = √(l(l+1) h/2π
For d orbital = √(2 x 3) h/2π = √6 h/2π
