Chemistry - Online Test
Q1. The
value of universal gas constant depends upon
Answer : Option D
Explaination / Solution:
No Explaination.
Q2. An
equilibrium constant of 3.2 × 10–6 for a reaction means, the
equilibrium is
Answer : Option B
Explaination / Solution:
Kc = [Products] / [Reactants]
3.2 x 10-6 = [Products] / [Reactants]
KC < 10-3 ; indicates
that [Reactant] >> [Product]
option (b) is correct,largely towards reverse direction.
Q3. The
heat of formation of CO and CO2 are – 26.4 kCal and – 94 kCal,
respectively. Heat of combus-tion of carbon monoxide will be
Answer : Option B
Explaination / Solution:
CO (g )+1/2O2 (g )→CO 2
(g)
ΔHC0 (CO) = ΔHf (CO2)
– ΔHf(CO) + ΔHf (O2)]
ΔHC0 (CO) = ΔHf (CO2)
– ΔHf(CO) + ΔHf (O2)]
ΔHC0 (CO) = –
94 KCal – [– 26.4 KCal + 0]
ΔHC0 (CO) = –
94 KCal + 26.4
KCal ΔHC0 (CO) = –
67.4 Kcal
Q4. Osometic pressure (p) of a solution is given by the relation
Answer : Option B
Explaination / Solution:
π = CRT
π = n/V . RT
π V = nRT
Q5. In
ClF3 ,NF3 and BF3 molecules the chlorine,
nitrogen and boron atoms are
Answer : Option D
Explaination / Solution:
ClF3 – Sp3d hybridisation
NF3 - Sp3 hybridisation
BF3 - Sp2 hybridisation
Q6. The
IUPAC name of the compound 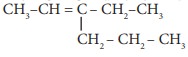 is
is
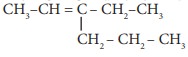 is
is
Answer : Option A
Explaination / Solution:
No Explaination.
Q7. Which of the following carbocation will be most stable?
Answer : Option D
Explaination / Solution:
No Explaination.
Q8. Hot
concentrated sulphuric acid is a moderately strong oxidising agent. Which of
the following reactions does not show oxidising behaviour?
Answer : Option C
Explaination / Solution:
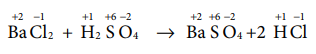
Q9. Which of the following compounds shall not produce propene by reaction with HBr followed by elemination (or) only direct elimination reaction
Answer : Option C
Explaination / Solution:
No Explaination.
Q10. The
electronic configuration of Eu (Atomic no. 63) Gd (Atomic no. 64) and Tb (Atomic
no. 65) are
Answer : Option B
Explaination / Solution:
Eu : [Xe] 4f7, 5d0, 6s2
Gd : [Xe] 4f7, 5d1, 6s2
Tb: [Xe] 4f9, 5d0, 6s2
