Chemistry - Online Test
Q1. The
IUPAC name of
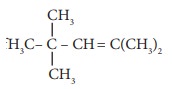
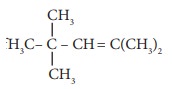
Answer : Option C
Explaination / Solution:
No Explaination.
Q2. -I effect is shown by
Answer : Option C
Explaination / Solution:
No Explaination.
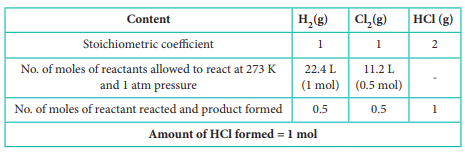
Q3. When
22.4 litres of H2 (g) is mixed with 11.2 litres of Cl2
(g), each at 273 K at 1 atm the moles of HCl (g), formed is equal to
Answer : Option D
Explaination / Solution:
H2(g) + Cl2(g) → 2 HCl (g)
Q4. The compound that will react most readily with gaseous bromine has the formula
Answer : Option A
Explaination / Solution:
No Explaination.
Q5. Two
electrons occupying the same orbital are distinguished by
Answer : Option B
Explaination / Solution:
Spin quantum number
For the first electron ms = +½
For the second electron ms = -½
Q6. The treatment of ethyl formate with excess of RMgX gives
Answer : Option C
Explaination / Solution:
No Explaination.
Q7. Which
one of the following arrangements represent the correct order of least negative
to most negative electron gain enthalpy
Answer : Option D
Explaination / Solution:
No Explaination.
Q8. Identify the wrong statement in the following
Answer : Option A
Explaination / Solution:
No Explaination.
Q9. The
hardness of water can be determined by volumetrically using the reagent
Answer : Option D
Explaination / Solution:
No Explaination.
Q10. Incase
of alkali metal halides, the ionic character increases in the order
Answer : Option B
Explaination / Solution:
ionic character (difference in electronegativity)
MI < MBr < MCl < MF
