Chemistry - Online Test
Q1. Which
of the following compounds will give racemic mixture on nucleophilic
substitution by OH- ion?
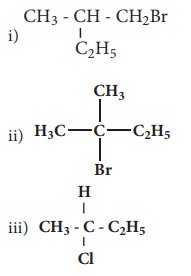

Answer : Option C
Explaination / Solution:
No Explaination.
Q2. Identify
the wrong statement.
Answer : Option A
Explaination / Solution:
No Explaination.
Q3. Ozone depletion will cause
Answer : Option C
Explaination / Solution:
No Explaination.
Q4. If
a body of a fish contains 1.2 g hydrogen in its total body mass, if all the
hydrogen is replaced with deuterium then the increase in body weight of the
fish will be
Answer : Option A
Explaination / Solution:
mass of deuterium = 2 × mass of protium
If all the 1.2 g hydrogen is replaced with
deuterium, the weight will become 2.4g. Hence the increase in body weight is
(2.4 – 1.2 = 1.2 g)
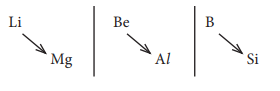
Q5. Lithium
shows diagonal relationship with
Answer : Option B
Explaination / Solution:
Q6. A
bottle of ammonia and a bottle of HCl connected through a long tube are opened
simultaneously at both ends. The white ammonium chloride ring first formed will
be
Answer : Option B
Explaination / Solution:
Rate of diffusion α 1/√m
mNH3 = 17 ; mHCl = 36.5
γNH3 > γHCl
Hence white fumes first formed near hydrogen
chloride
Q7.
In the equilibrium,
2A(g) ⇌ 2B(g) + C2(g)
the equilibrium concentrations of A, B and C2 at
400 K are 1 × 10–4 M, 2.0 × 10–3 M, 1.5 × 10–4
M respectively. The value of Kc for the equilibrium at 400 K is
Answer : Option A
Explaination / Solution:
[A] = 1 × 10–4
M ;
[B] = 2 × 10–3 M
[C] = 1.5 × 10–4 M
2A (g) ⇌ 2B(g) + C2(g)
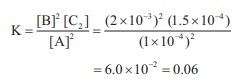
Q8. Heat
of combustion is always
Answer : Option B
Explaination / Solution:
No Explaination.
Q9. P1
and P2 are the vapour pressures of pure liquid components, 1 and 2
respectively of an ideal binary solution if x1 represents the mole fraction
of component 1, the total pressure of the solution formed by 1 and 2 will be
Answer : Option C
Explaination / Solution:
Ptotal = P1 + P2
= P1x1 + P2x2 (x1 + x2 = 1)
= P1 (1 – x2) + P2x2
(x1 = 1 – x2)
= P1 – P1x2 + P2x2
= P1 – x2 (P1 – P2)
Q10. According to Valence bond theory, a bond between two atoms is formed when
Answer : Option B
Explaination / Solution:
two half filled orbitals overlap.
