Chemistry - Online Test
Q1. In
a closed room of 1000 m3 a perfume bottle is opened up. The room develops a
smell. This is due to which property of gases?
Answer : Option C
Explaination / Solution:
No Explaination.
Q2.
K1 and K2 are the equilibrium
constants for the reactions respectively.
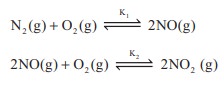
What is the equilibrium constant for the reaction NO2(g)
⇌ ½N2(g) + O2(g)
Answer : Option A
Explaination / Solution:


Q3. An
ideal gas expands from the volume of 1 × 10–3 m3 to 1 ×
10–2 m3 at 300 K against a constant pressure at 1 × 105
Nm–2. The work done is
Answer : Option A
Explaination / Solution:
w = – P ∆ V
w = – (1 × 105 Nm–2)
(1× 10–2 m3 – 1 × 10–3
m3)
w= –105 (10–2 – 10–3)
Nm
w = – 105 (10 – 1) 10–3) J
w = – 105 (9 × 10–3) J
w = –9 × 102 J w = –900 J
Q4. Which one of the following gases has the lowest value of Henry's law constant ?
Answer : Option C
Explaination / Solution:
Carbon dioxide ; most stable gas and has lowest value of Henrys Law constant.
Q5.
Assertion: Oxygen
molecule is paramagnetic.
Reason : It has two
unpaired electron in its bonding molecular orbital
Answer : Option C
Explaination / Solution:
Correct Statement :
Oxygen molecule is paramagnetic Correct Reason
It has two unpaired electron in its antibond-ing molecular
orbital.
Option (c) assertion true and reason false.
Q6. IUPAC
name of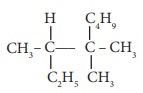

Answer : Option C
Explaination / Solution:
No Explaination.
Q7. Which of the following species does not exert a resonance effect?
Answer : Option D
Explaination / Solution:
No Explaination.
Q8. When
6.3 g of sodium bicarbonate is added to 30 g of acetic acid solution, the
residual solution is found to weigh 33 g. The number of moles of carbon dioxide
released in the reaction is
Answer : Option C
Explaination / Solution:
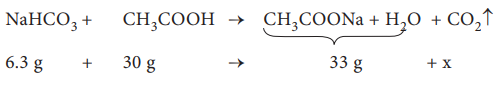
The amount of CO2 released, x = 3.3 g
No. of moles of CO2 released = 3.3 / 44
= 0.075 mol
Q9. The general formula for cyclo alkanes
Answer : Option B
Explaination / Solution:
No Explaination.
Q10. Which
of the following pairs of d-orbitals will have electron density along the axes
?
Answer : Option C
Explaination / Solution:
No Explaination.
