Chemistry - Online Test
Q1. The
IUPAC name of the compound CH3–CH= CH – C ≡ CH is
Answer : Option B
Explaination / Solution:
No Explaination.
Q2. Which of the group has highest +I effect?
Answer : Option D
Explaination / Solution:
No Explaination.
Q3. 1 g
of an impure sample of magnesium carbonate (containing no thermally
decomposable impurities) on complete thermal decomposition gave 0.44 g of carbon
dioxide gas. The percentage of impurity in the sample is
Answer : Option C
Explaination / Solution:
No Explaination.
Q4. The compounds formed at anode in the electrolysis of an aquous solution of potassium acetate are
Answer : Option C
Explaination / Solution:
No Explaination.
Q5.
Assertion : The spectrum of He+ is expected
to be similar to that of hydrogen
Answer : Option A
Explaination / Solution:
No Explaination.
Q6. In
the reaction
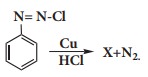

X is _________
Answer : Option B
Explaination / Solution:
No Explaination.
Q7. In
the third period the first ionization potential is of the order.
Answer : Option B
Explaination / Solution:
No Explaination.
Q8. The pH of normal rain water is
Answer : Option D
Explaination / Solution:
No Explaination.
Q9.
Assertion : Permanent hardness of water is removed
by treatment with washing soda.
Answer : Option A
Explaination / Solution:
Ca2+
+ Na2CO3 → CaCO3 ↓ + 2Na+
Q10. Find
the wrong statement
Answer : Option C
Explaination / Solution:
Potassium carbonate cannot be prepared by solvay
process. Potassium bicarbonate is fairly soluble in water and does not
precipitate out.
