Chemistry - Online Test
Q1. The correct difference between first and second order reactions is that
Answer : Option B
Explaination / Solution:
For a first order
reaction
t1/2 =
0.6932/k
For a second order
reaction
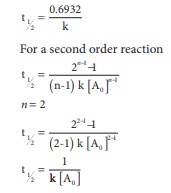
Q2. Which of the following reduction is not thermodynamically feasible?
Answer : Option B
Explaination / Solution:
No Explaination.
Q3.
Cell equation : A + 2B- → A2+
+2B;
Answer : Option A
Explaination / Solution:
No Explaination.
Q4. Which one of the following is not correct?
Answer : Option A
Explaination / Solution:
No Explaination.
Q5. The
major product of the following reaction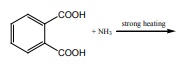

Answer : Option B
Explaination / Solution:
No Explaination.
Q6. A two dimensional solid
pattern formed by two different atoms X and Y is shown below. The black and
white squares represent atoms X and Y respectively. the simplest formula for
the compound based on the unit cell from the pattern is
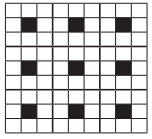

Answer : Option A
Explaination / Solution:
No Explaination.
Q7. Which of the following amino acids are achiral?
Answer : Option D
Explaination / Solution:
No Explaination.
Q8. After 2 hours, a
radioactive substance becomes (1/16)th of original amount. Then the
half life ( in min) is
Answer : Option C
Explaination / Solution:
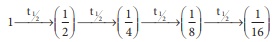
4t1/2 = 2
hours
t1/2 = 30 min
Q9. Which of the following is not true with respect to Ellingham diagram?
Answer : Option B
Explaination / Solution:
No Explaination.
