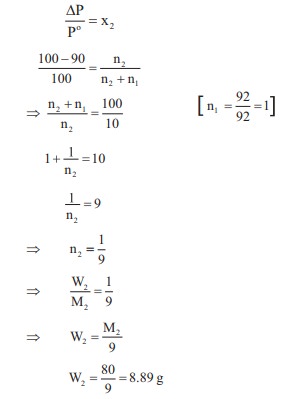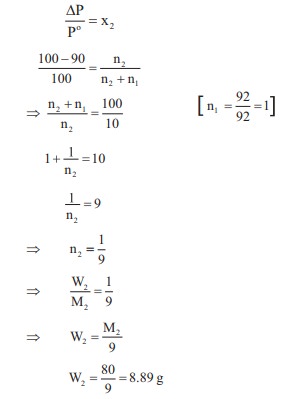UNIT 9: Solutions - Online Test
Q1. The Henry's law constants for two gases A and B are x and y respectively. The ratio of mole fractions of A to B is 0.2. The ratio of mole fraction of B and A dissolved in water will be
Answer : Option D
Explaination / Solution:
Given, (KH)A = x
(KH)B = y
xA/xB = 0.2
(xA/xB)in solution = ?
PA = x (xA) in solution –
(1)
PB = y (xB) in solution –
(2)
(xA/xB)in solution = (PB/PA)
. (x/y) = (xB/xA) . (x/y) = (1/0.2) . (x/y) = 5x/y
Q2. At
100oC the vapour pressure of a solution containing 6.5g a solute in
100g water is 732mm. If Kb = 0.52, the boiling point of this solution
will be
Answer : Option C
Explaination / Solution:

Tb – 100 = 1.06
Tb = 100 + 1.06
= 101.06 ≈ 101o C
Q3. According to Raoults law, the relative lowering of vapour pressure for a solution is equal to
Answer : Option B
Explaination / Solution:
∆P/Pº=
x2 (mole fraction of the solute)
Q4. At same temperature, which pair of the following solutions are isotonic ?
Answer : Option D
Explaination / Solution:
0.1 × 3 ion [Ba2+,
2NO3–]
= 0.1 × 3 ion [2 Na+,
SO4–]
Q5. The
empirical formula of a non-electrolyte(X) is CH2O. A solution
containing six gram of X exerts the same osmotic pressure as that of 0.025M
glucose solution at the same temperature. The molecular formula of X is
Answer : Option B
Explaination / Solution:
(π1)non electrolyte = (π2)glucose
C1RT = C2RT
[CH2O
=> 12+2+16=30]
W1/M1 = W1/M2
6 / n(30) = 0.025
n = 8
∴ molecular
formula = C8H16O8
Q6. The
KH for the solution of oxygen dissolved in water is 4 × 104
atm at a given temperature. If the partial pressure of oxygen in air is 0.4
atm, the mole fraction of oxygen in solution is
Answer : Option C
Explaination / Solution:


Q7. Normality of 1.25M sulphuric acid is
Answer : Option C
Explaination / Solution:
Normality of H2SO4 = (no.of replacable
H+)×M
= 2 × 1.25
= 2.5 N
Q8. Two liquids X and Y on mixing gives a warm solution. The solution is
Answer : Option D
Explaination / Solution:
ΔHmix
is negative and show negative deviation from Raoults law.
Q9. The
relative lowering of vapour pressure of a sugar solution in water is 3.5 × 10–3.
The mole fraction of water in that solution is
Answer : Option D
Explaination / Solution:
∆P / Pº =
Xsugar
3.5 × 10–3 = Xsugar
Xsugar + XH2o = 1
XH2o = 1 –
0.0035 = 0.9965
Q10. The
mass of a non-voltaile solute (molar mass 80 g mol–1) which should
be dissolved in 92g of toluene to reduce its vapour pressure to 90%
Answer : Option D
Explaination / Solution: