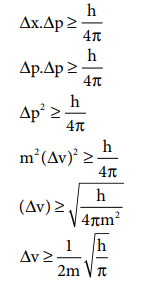UNIT 2: Quantum Mechanical Model of Atom - Online Test
Orbital angular momentum = √(l(l+1) h/2π
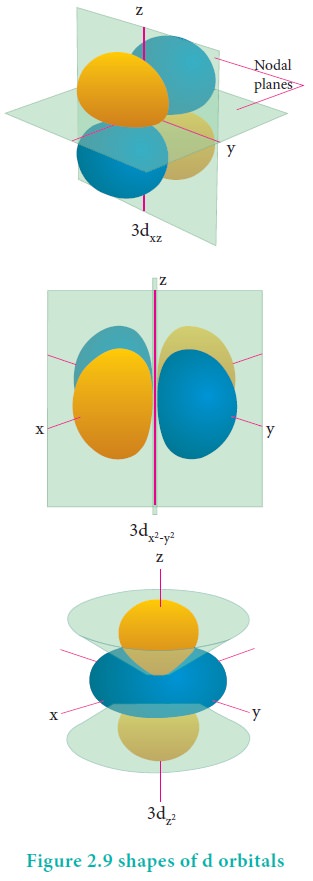
For d orbital = √(2 x 3) h/2π = √6 h/2π
n = 3; l=1; m = -1
either 3px or 3py
i.e., Maximum two electrons can be accomodated
either in 3px or in 3py
Assertion : Number of radial and angular nodes for
3p orbital are 1, 1 respectively.
No. of radial node = n-l-1
No. of angular node = l
for 3p orbital
No. of angular node = l = 1
No. of radial node = n-l-1 = 3-1-1 = 1
n = 3; l = 0; ml = 0 - one s orbital
n = 3; l = 1; ml = -1, 0, 1 - three p
orbitals
n = 3; l = 2; ml = -2, -1, 0, 1, 2 -
five d Orbitals
Overall nine orbitals are possible.
n = 6
According Aufbau principle,
6s →4f →5d →6p
ns → (n-1)f →(n-2)d → np

(ii) l
can have the values from 0 to n-1
n=2; possible 'l' values are 0, 1 hence l = 2 is not possible.
(iv)for l=0;
m=-1 not possible
(v)for n=3 l
= 4 and m = 3 not possible
n+l = 8
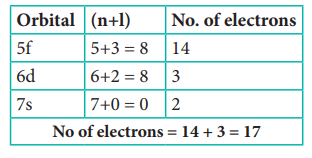
Electronic configuration of atom with atomic number
105 is [Rn] 5f14 6d3 7s2