Electrical Engineering - Online Test
Q1. The reaction rate involving reactants A and B is given by 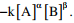 Which one of the
following statements is valid for the reaction to be first –order reaction?
Which one of the
following statements is valid for the reaction to be first –order reaction?
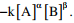 Which one of the
following statements is valid for the reaction to be first –order reaction?
Which one of the
following statements is valid for the reaction to be first –order reaction?
Answer : Option B
Explaination / Solution:
In chemical kinetics, the order of reaction with respect to given substance is defined as the
index or exponent to which its concentration term in the rate equation is raised.

Order of reaction = α + β
For first order reaction, α + β = 1
Q2. The matrix P is the inverse of a matrix Q. If I denotes the identity matrix, which one of the
following options is correct?
Answer : Option C
Explaination / Solution:
Given P is inverse of Q
⇒ PQ = QP = I
Q3. Let x be a continuous variable defined over the interval (−∞, ∞), and 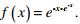
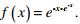
Answer : Option B
Explaination / Solution:
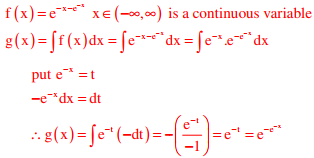
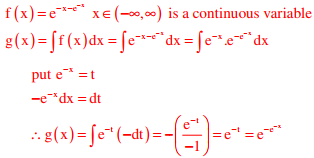
Q4. For the function f(x) = a + bx, 0 ≤ x ≤ 1, to be a valid probability density function, which one of the following statements is correct?
Answer : Option B
Explaination / Solution:
f(x) = a + bx 0 ≤ x ≤1 iis a valid probability density function
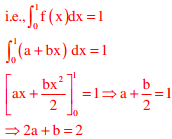
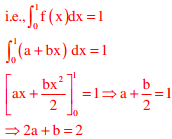
a=0.5, b=1 satisfies the above relation
Q5. Consider the matrix 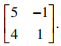 Which one of the following statements is TRUE for the
eigenvalues and eigenvectors of this matrix?
Which one of the following statements is TRUE for the
eigenvalues and eigenvectors of this matrix?
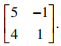 Which one of the following statements is TRUE for the
eigenvalues and eigenvectors of this matrix?
Which one of the following statements is TRUE for the
eigenvalues and eigenvectors of this matrix?
Answer : Option A
Explaination / Solution:
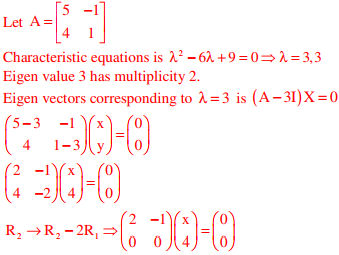
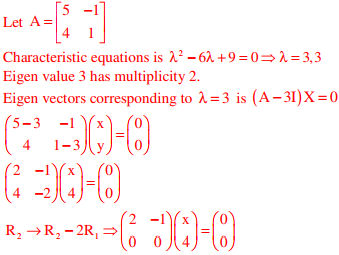
e(A) = 1
Number of linearly independent eigen vectors corresponding to eigen value λ = 3 is
n-r=2-1=1 where n= no. of unknowns, r= rank of( A − λI)
One linearly independent eigen vector exists corresponding to λ = 3
Q6. The solution of the equation (dQ/dt) + Q = 1 with Q = 0 at t = 0 is
Answer : Option D
Explaination / Solution:
(dQ/dt) + Q = 1 and Q = 0 at t = 0
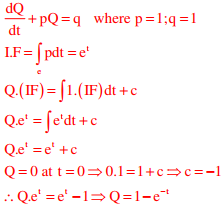
Comparing with first order linear differential equations
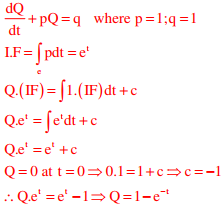
Q7. The following sequence of numbers is arranged in increasing order: 1, x, x, x, y, y,9,16,18.
Given that the mean and median are equal, and are also equal to twice the mode, the value of
y is
Answer : Option D
Explaination / Solution:
Given, Mean = Median = 2Mode
Mean = Median = 2x [Mode = x].............. (1)
Mean of the data = (3x + 2y + 44)/9
2x = (3x + 2y + 44)/9
15x - 2y = 44.............(2)
Median of the data = y.........(3)
y = 2x ..........(4)
From (2); 11x = 44
x = 4
y = 8
Q8. If the radius of a right circular cone is increased by 50%, its volume increases by
Answer : Option C
Explaination / Solution:
Given, radius of a right circular cone is increased by 50%. 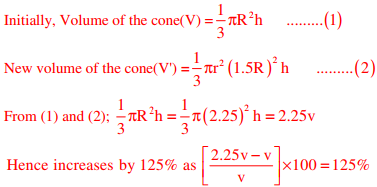
Let, height of the circular cone=(h)
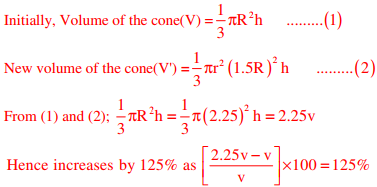
Q9. In a process, the number of cycles to failure decreases exponentially with an increase
in load. At a load of 80 units, it takes 100 cycles for failure. When the load is halved, it
takes 10000 cycles for failure. The load for which the failure will happen in 5000
cycles is __________.
Answer : Option B
Explaination / Solution:
From the data given we assume
load = exp onent/log(cycles)
80 = x/log(10000)
x = 160
40 = x/log(10000)
x = 160
load = 160/log 5000 = 43.25
Q10. If f(x7) = 2x7 + 3x - 5, which of the following is a factor of f(x)?
Answer : Option B
Explaination / Solution:
from the option (b0 substitute x=1 in
2x7 + 3x - 5 = 0
2(1)7 + 3(1) - 5 = 0
5 - 5 = 0
So (x - 1) is a factor of f (x)
