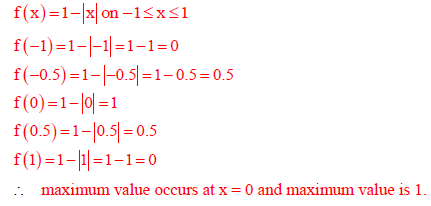Mechanical Engineering - Online Test
Q1. For the stability of a floating body the
Answer : Option D
Explaination / Solution:
No Explaination.
Q2. To evaluate the double integral 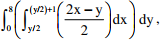 we make the substitution
we make the substitution 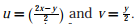 The integral will reduce to
The integral will reduce to
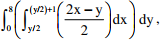 we make the substitution
we make the substitution 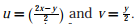 The integral will reduce to
The integral will reduce to
Answer : Option B
Explaination / Solution:


Q3. For the four-bar linkage shown in the figure, the angular velocity of link AB is 1
rad/s. the length of link CD is 1.5 times the length of link AB. In the configuration
shown, the angular velocity of link CD in rad/s is
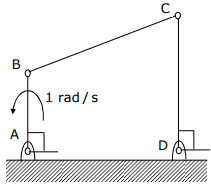

Answer : Option D
Explaination / Solution:
For the given configuration
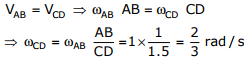
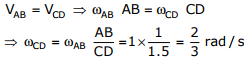
Q4. In an experimental set-up, air flows between two stations P and Q adiabatically.
The direction of flow depends on the pressure and temperature conditions
maintained at P and Q. The conditions at station P are 150kPa and 350K. The
temperature at station Q is 300K.
The following are the properties and relations pertaining to air:
Specific heat at constant pressure, Cp = 1.005kJ / kgK;
Specific heat at constant volume, Cv = 0.718kJ / kgK;
Characteristic gas constant, R = 0.287kJ / kgK
Enthalpy, h = CpT
Internal energy, u = CvT
If the air has to flow from station P to station Q, the maximum possible value of
pressure in kPa at station Q is close to
Answer : Option B
Explaination / Solution:
To cause the flow from P to Q, change in entropy (SP - SQ) should be
greater than zero 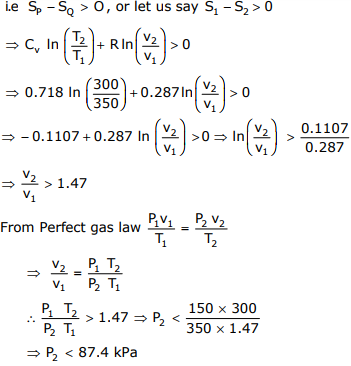
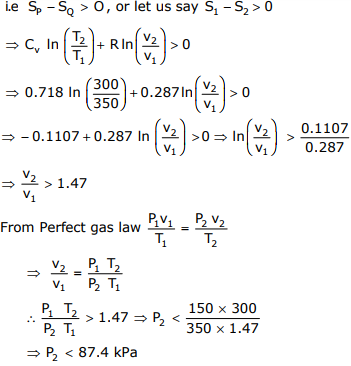
∴ The maximum value of pressure at Q = 87 kPa
Q5. The crystal structure of aluminium is
Answer : Option B
Explaination / Solution:
No Explaination.
Q6. The minimum value of the function f(x) = x3 - 3x2 - 24x +
100 in the interval [-3. 3] is
Answer : Option B
Explaination / Solution:
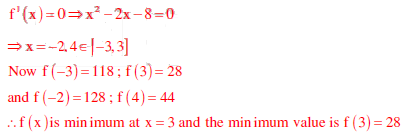
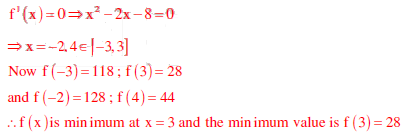
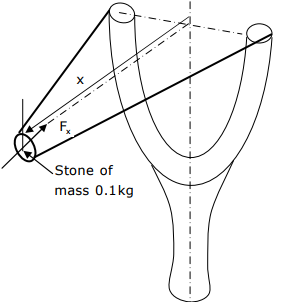
Q7. A stone with mass of 0.1kg is catapulted as shown in the figure. The total force Fx (in N) exerted by the rubber band as a function of distance x (in m) is given by F = 300x2. If the stone is displaced by 0.1m from the un-stretched position
(x=0) of the rubber band, the energy stored in the rubber band is

Answer : Option B
Explaination / Solution:
Energy stored in the bar = W.D. by the stone
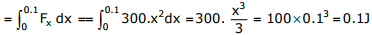
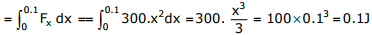
Q8. In an experimental set-up, air flows between two stations P and Q adiabatically. The direction of flow depends on the pressure and temperature conditions maintained at P and Q. The conditions at station P are 150kPa and 350K. The temperature at station Q is 300K.
The following are the properties and relations pertaining to air:
Specific heat at constant pressure, Cp = 1.005kJ / kgK;
Specific heat at constant volume, Cv = 0.718kJ / kgK;
Characteristic gas constant, R = 0.287kJ / kgK
Enthalpy, h = CpT
Internal energy, u = CvT
If the pressure at station Q is 50kPa, the change in entropy (SQ - SP) in kJ/kgK is
Answer : Option C
Explaination / Solution:
No Explaination.
Q9. The “Jominy test” is used to find
Answer : Option B
Explaination / Solution:
Jominy Test measures the hardenability of the steels.
Q10. Consider a function f(x) = 1 - |x| on -1 ≤ x ≤ 1. The value of x at which the function attains a maximum, and the maximum value of function are:
Answer : Option C
Explaination / Solution: