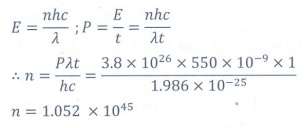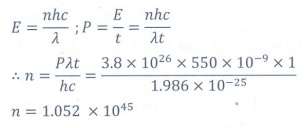UNIT VIII: Dual Nature of Radiation and Matter - Online Test
Q1. The wavelength λe of an electron and
λp of a photon of same energy E are related by
Answer : Option D
Explaination / Solution:
de
Broglie wavelength for an electron
λe=
h / √(2mE) ;
λe ∝ 1/√E ;
λe2 ∝ 1/E ------------- (1)
For
photon
λp=
hc / E ;
λp ∝ 1/E ------------- (2)
From
equation (1) and (2), we have So,
λp ∝ λe2
Q2. In an electron microscope, the electrons are accelerated by a voltage of 14 kV. If the voltage is changed to 224 kV, then the de Broglie wavelength associated with the electrons would
Answer : Option C
Explaination / Solution:
deBroglie
wavelength λ ∝ 1/√V
λ1/λ2 = √(V2/V1)
= √(224/14) = √16 = 4
λ1
= 4λ2
λ2
= λ1/4
Q3. A particle of mass 3 × 10–6 g has the
same wavelength as an electron moving with a velocity 6×106 m s−1
. The velocity of the particle is
Answer : Option D
Explaination / Solution:
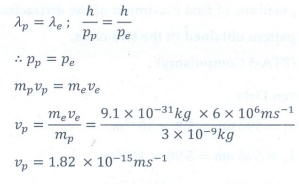
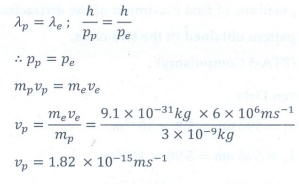
Q4. When a metallic surface is illuminated with radiation of wavelength λ, the stopping potential is V. If the same surface is illuminated with radiation of wavelength 2λ, the stopping potential is V/4. The threshold wavelength for the metallic surface is
Answer : Option D
Explaination / Solution:
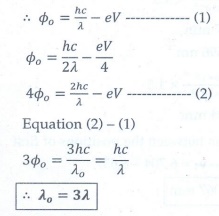
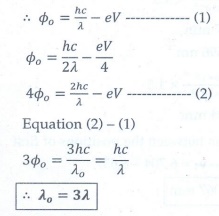
Q5. If a light of wavelength 330 nm is incident on a metal with work
function 3.55 eV, the electrons are emitted. Then the wavelength of the emitted
electron is (Take h = 6.6 × 10–34 Js)
Answer : Option B
Explaination / Solution:
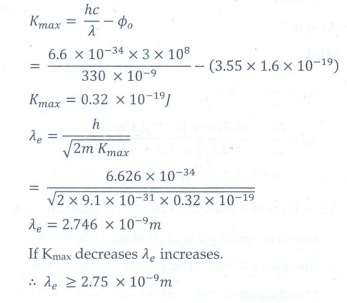
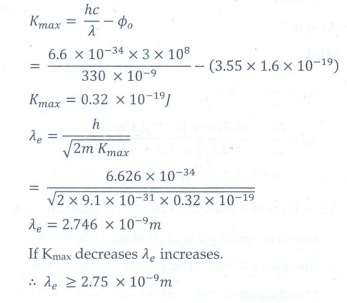
Q6. A photoelectric surface is illuminated successively by monochromatic light of wavelength λ and λ/2. If the maximum kinetic energy of the emitted photoelectrons in the second case is 3 times that in the first case, the work function at the surface of material is
Answer : Option D
Explaination / Solution:
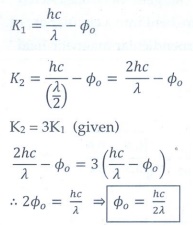
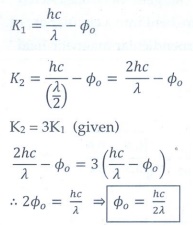
Q7. In photoelectric emission, a radiation whose frequency is 4 times threshold frequency of a certain metal is incident on the metal. Then the maximum possible velocity of the emitted electron will be
Answer : Option B
Explaination / Solution:
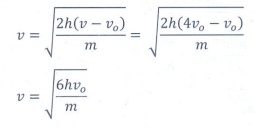
From
Enistein's equation
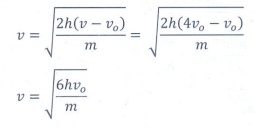
Q8. Two radiations with photon energies 0.9 eV and 3.3 eV respectively are falling on a metallic surface successively. If the work function of the metal is 0.6 eV, then the ratio of maximum speeds of emitted electrons will be
Answer : Option B
Explaination / Solution:


Q9. A light source of wavelength 520 nm emits 1.04 ×
1015 photons per second while the second source of 460 nm produces 1.38 × 1015 photons per
second. Then the ratio of power of second source to that of first source is
Answer : Option C
Explaination / Solution:
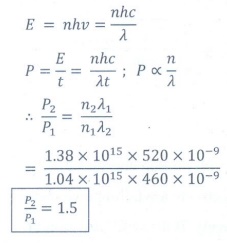
Q10. The mean wavelength of light from sun is taken
to be 550 nm and its mean power is 3.8 × 1026W. The number of
photons received by the human eye per second on the average from sunlight is of
the order of
Answer : Option A
Explaination / Solution: