Thermodynamics - Online Test
Q1. For the reaction, , what are the signs of and ?
Answer : Option B
Explaination / Solution:
As in this cas e bond formation between two Cl atom is taking place so energy will be released so ΔH<0 and since 2 atoms are combining to form 1 molecule randomness decreases so ΔS<0.
Q2.
for the reaction . Calculate for the reaction, and predict whether the reaction may occur spontaneously.
Answer : Option C
Explaination / Solution:
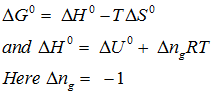
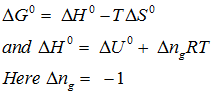
Q3. The equilibrium constant for a reaction is 10. What will be the value of , T = 300 K.
Answer : Option A
Explaination / Solution:
= -RTln K
Q4. Calculate the heat and the work associated with a process in which 5.00 mol of gas expands reversibly at constant temperature T = 298 K from a pressure of 10.00 to 1.00 atm
Answer : Option B
Explaination / Solution:
for isothermal process ΔU=0 while –w=nRTln(P1/P2) = q
Q5. Thermodynamics is not concerned about
Answer : Option B
Explaination / Solution:
Thermodynamics on tells that whether reaction will give take place or not. It doesnot tell the rate at which reaction will occur.
Q6. Which of the following statements is correct?
Answer : Option D
Explaination / Solution:
It will be a closed system.
Q7. The state of a gas can be described by quoting the relationship between
Answer : Option C
Explaination / Solution:
State of gas is determined by stating the value of P,V,n,T
Q8. The volume of gas is reduced to half from its original volume. The specific heat will be
Answer : Option C
Explaination / Solution:
Specific heat will remain constant.
Q9. The enthalpies of elements in their standard states are taken as zero. The enthalpy of formation of a compound
Answer : Option C
Explaination / Solution:
Enthapy of formation of a compound from elements in standard state can b +ve or –ve.
Q10. Enthalpy of sublimation of a substance is equal to
Answer : Option A
Explaination / Solution:
The process of sublimation involves the change of solid into vapour. Though in sublimation a solid does not pass through the liquid phase on its way to the gas phase, the enthalpy change is equal to the sum of enthalpy of fusion and enthalpy of vaporization because enthalpy is a state function.
