Thermodynamics - Online Test
Q1. A reaction, A + B → C + D + q is found to have a positive entropy change. The reaction will be
Answer : Option C
Explaination / Solution:
ΔG = ΔH -T ΔS
ΔS is positive and ΔH is negative as heat is liberated in the reaction.
so ΔG is negative hence reaction will be spontaneous at all temperature.
Q2. In a process, 701 J of heat is absorbed by a system and 394 J of work is done by the system. What is the change in internal energy for the process?
Answer : Option C
Explaination / Solution:
ΔU = q + w (1st law of thermodynamics)
q = +701J (heat is absorbed by system)
w = -394 J (work is done by system)
ΔU = 701- 394
= 307J
Q3. The reaction of cyanamide, NCN (s), with dioxygen was carried out in a bomb calorimeter, and was found to be 742.7 kJ at 298 K. Calculate enthalpy change for the reaction at 298 K. NCN(g) +
Answer : Option B
Explaination / Solution:
RT where Δng = 2-((3/2)+1)
Q4. Calculate the number of kJ of heat necessary to raise the temperature of 60.0 g of aluminium from 35 to 55. Molar heat capacity of Al is 24 J
Answer : Option D
Explaination / Solution:
q=nC ΔT where n= 60.0/27 mole and ΔT = 20 K and C = 24J/mol K
Q5. Calculate the enthalpy change on freezing of 1.0 mol of water at 10.0 to ice at -10.0. H = 6.03 kJ at 0.1
Answer : Option C
Explaination / Solution:
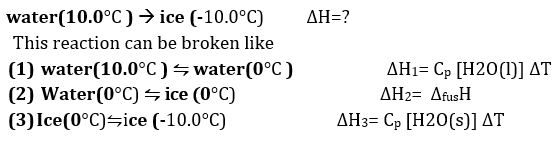

Q6.
Enthalpy of combustion of carbon to is –393.5 kJ . Calculate the heat released upon formation of 35.2 g of from carbon and dioxygen gas.
Answer : Option A
Explaination / Solution:
when 1 mole of is produced energy released is –393.5 kJ Moles of given =35.2/44 =0.8 moles So energy released = 0.8 x393.5 KJ/mol = 315 KJ/mol
Q7. Enthalpies of formation of CO(g), , and are -110, –-393, 81 and 9.7 kJ respectively. Find the value of for the reaction:
Answer : Option C
Explaination / Solution:
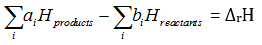
Q8. Given = –-92.4 kJ What is the standard enthalpy of formation of gas?
Answer : Option B
Explaination / Solution:
Standard Enthalpy of formation of NH3 is the energy change that take place when 1 mole of NH3 is formed from elements in standard state. For 2 moles of ammonia formation energy= –-92.4 kJ . For 1 mole energy =0.5 x–-92.4 kJ = -46.2 kJ
Q9. For an isolated system, ΔU = 0, what will be ΔS?
Answer : Option A
Explaination / Solution:
for isolated system ΔS = 0.
Q10. for the reaction at 298 K, = 400 kJ and = 0.2 kJ At what temperature will the reaction become spontaneous considering and to be constant over the temperature range.
Answer : Option B
Explaination / Solution:
ΔG= ΔH-TΔS At equilibrium ΔG=0 then T= ΔH/ΔS =2000K
