UNIT VIII: Heat and Thermodynamics - Online Test
Q1. In hot summer after a bath, the body’s
Answer : Option A
Explaination / Solution:
No Explaination.
Q2. The graph between volume and temperature in Charles’ law is
Answer : Option C
Explaination / Solution:
No Explaination.
Q3. When a cycle tyre suddenly bursts, the air inside the tyre expands. This process is
Answer : Option B
Explaination / Solution:
No Explaination.
Q4. An
ideal gas passes from one equilibrium state (P1, V1,
T1, N) to another equilibrium state (2P1, 3V1,
T2, N). Then
Answer : Option B
Explaination / Solution:
No Explaination.
Q5. When a uniform rod is heated, which of the following quantity of the rod will increase
Answer : Option D
Explaination / Solution:
No Explaination.
Q6. When food is cooked in a vessel by keeping the lid closed, after some time the steam pushes the lid outward. By considering the steam as a thermodynamic system, then in the cooking process
Answer : Option A
Explaination / Solution:
No Explaination.
Q7. When you exercise in the morning, by considering your body as thermodynamic system, which of the following is true?
Answer : Option B
Explaination / Solution:
No Explaination.
Q8. A hot cup of coffee is kept on the table. After some time it attains a thermal equilibrium with the surroundings. By considering the air molecules in the room as a thermodynamic system, which of the following is true
Answer : Option C
Explaination / Solution:
No Explaination.
Q9. An
ideal gas is taken from (Pi,Vi) to (Pf,Vf)
in three different ways. Identify the process in which the work done on the gas
the most.
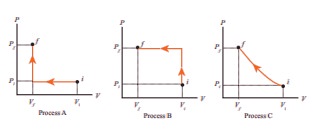

Answer : Option B
Explaination / Solution:
No Explaination.
Q10. The
V-T diagram of an ideal gas which goes through a reversible cycle A→B→C→D is
shown below. (Processes D→A and B→C are adiabatic)
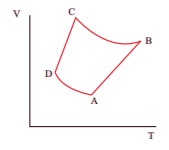

The
corresponding PV diagram for the process is (all figures are schematic)
Answer : Option B
Explaination / Solution:
No Explaination.
