Structure of Atom - Online Test
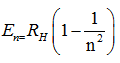
where n=1,2,3....... . In this equation, is called
he commonly occurring isotopes of carbon atoms are: 612C, 613C, 614C . They contain:
Isotopes are atoms with the same number of protons but that have a different number of neutrons.
Atomic number of carbon = 6
So number of protons in each isotope = 6
Number of neutrons in = 12-6= 6
Number of neutrons in = 13-6= 7
Number of neutrons in = 14-6= 8
How many protons, neutrons and electrons are in ?
Atomic number of iron (Fe) = 26
So number of protons = 26
Number of protons = number of electrons
But To gain 3+ charge it should loose 3 electron. So number of electrons present = 26-3 = 23
Now atomic mass = 56 (Given).
Number of neutrons = atomic mass - number of protons = 56-26 = 30
Give the number of electrons in the species, and .
atomic number O has atomic number = 8 so number of electrons in = 16
while in there is one unit positive charge so no. of electron =15.
